Segmental patterns of vestibular-mediated synaptic inputs to axial and limb motoneurons in the neonatal mouse assessed by optical recording
- PMID: 20962007
- PMCID: PMC3036187
- DOI: 10.1113/jphysiol.2010.195644
Segmental patterns of vestibular-mediated synaptic inputs to axial and limb motoneurons in the neonatal mouse assessed by optical recording
Abstract
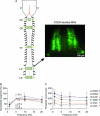
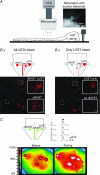
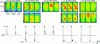
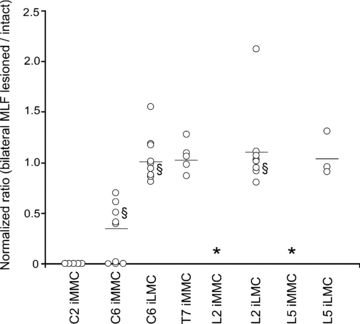
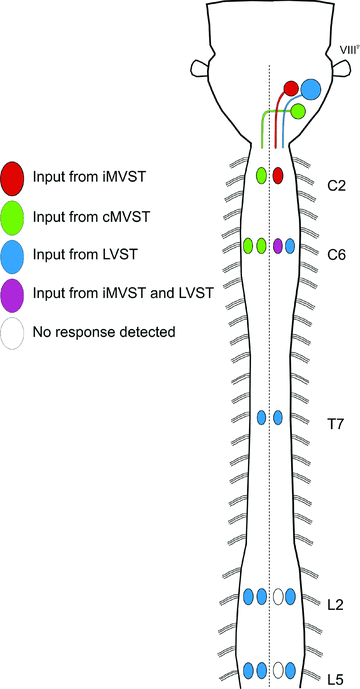
Proper control of movement and posture occurs partly via descending projections from the vestibular nuclei to spinal motor circuits. Days before birth in rodents, vestibulospinal neurons develop axonal projections that extend to the spinal cord. How functional these projections are just after birth is unknown. Our goal was to assess the overall functional organization of vestibulospinal inputs to spinal motoneurons in a brainstem-spinal cord preparation of the neonatal mouse (postnatal day (P) 0-5). Using calcium imaging, we recorded responses evoked by electrical stimulation of the VIIIth nerve, in many motoneurons simultaneously throughout the spinal cord (C2, C6, T7, L2 and L5 segments), in the medial and lateral motor columns. Selective lesions in the brainstem and/or spinal cord distinguished which tracts contributed to the responses: those in the cervical cord originated primarily from the medial vestibulospinal tracts but with a substantial contribution from the lateral vestibulospinal tract; those in the thoracolumbar cord originated exclusively from the lateral vestibulospinal tract. In the thoracolumbar but not the cervical cord, excitatory commissural connections mediated vestibular responses in contralateral motoneurons. Pharmacological blockade of GABA(A) receptors showed that responses involved a convergence of excitatory and inhibitory inputs which in combination produced temporal response patterns specific for different segmental levels. Our results show that by birth vestibulospinal projections in rodents have already established functional synapses and are organized to differentially regulate activity in neck and limb motoneurons in a tract- and segment-specific pattern similar to that in adult mammals. Thus, this particular set of descending projections develops several key features of connectivity appropriately at prenatal stages. We also present novel information about vestibulospinal inputs to axial motoneurons in mammals, providing a more comprehensive platform for future studies into the overall organization of vestibulospinal inputs and their role in regulating postural stability.
Figures





Similar articles
-
Early postnatal maturation in vestibulospinal pathways involved in neck and forelimb motor control.Dev Neurobiol. 2016 Oct;76(10):1061-77. doi: 10.1002/dneu.22375. Epub 2016 Jan 22. Dev Neurobiol. 2016. PMID: 26724676
-
Vestibular-mediated synaptic inputs and pathways to sympathetic preganglionic neurons in the neonatal mouse.J Physiol. 2012 Nov 15;590(22):5809-26. doi: 10.1113/jphysiol.2012.234609. Epub 2012 Sep 3. J Physiol. 2012. PMID: 22946097 Free PMC article.
-
Segmental organization of vestibulospinal inputs to spinal interneurons mediating crossed activation of thoracolumbar motoneurons in the neonatal mouse.J Neurosci. 2015 May 27;35(21):8158-69. doi: 10.1523/JNEUROSCI.5188-14.2015. J Neurosci. 2015. PMID: 26019332 Free PMC article.
-
Development and regenerative capacity of descending supraspinal pathways in tetrapods: a comparative approach.Adv Anat Embryol Cell Biol. 2000;154:iii-ix, 1-145. doi: 10.1007/978-3-642-57125-1. Adv Anat Embryol Cell Biol. 2000. PMID: 10692782 Review.
-
Descending Influences on Vestibulospinal and Vestibulosympathetic Reflexes.Front Neurol. 2017 Mar 27;8:112. doi: 10.3389/fneur.2017.00112. eCollection 2017. Front Neurol. 2017. PMID: 28396651 Free PMC article. Review.
Cited by
-
The Larval Zebrafish Vestibular System Is a Promising Model to Understand the Role of Myelin in Neural Circuits.Front Neurosci. 2022 May 6;16:904765. doi: 10.3389/fnins.2022.904765. eCollection 2022. Front Neurosci. 2022. PMID: 35600621 Free PMC article.
-
Catecholaminergic dysfunction drives postural and locomotor deficits in a mouse model of spinal muscular atrophy.Cell Rep. 2025 Jan 28;44(1):115147. doi: 10.1016/j.celrep.2024.115147. Epub 2025 Jan 2. Cell Rep. 2025. PMID: 39752251 Free PMC article.
-
Brainstem Steering of Locomotor Activity in the Newborn Rat.J Neurosci. 2018 Aug 29;38(35):7725-7740. doi: 10.1523/JNEUROSCI.1074-18.2018. Epub 2018 Jul 23. J Neurosci. 2018. PMID: 30037828 Free PMC article.
-
Retrograde loading of nerves, tracts, and spinal roots with fluorescent dyes.J Vis Exp. 2012 Apr 19;(62):4008. doi: 10.3791/4008. J Vis Exp. 2012. PMID: 22547001 Free PMC article.
-
Altered neurofilament protein expression in the lateral vestibular nucleus in Parkinson's disease.Exp Brain Res. 2017 Dec;235(12):3695-3708. doi: 10.1007/s00221-017-5092-3. Epub 2017 Sep 19. Exp Brain Res. 2017. PMID: 28929183
References
-
- Akaike T, Fanardjian VV, Ito M, Ono T. Electrophysiological analysis of the vestibulospinal reflex pathway of rabbit. II. Synaptic actions upon spinal neurones. Exp Brain Res. 1973;17:497–515. - PubMed
-
- Altman J, Bayer SA. Development of the brain stem in the rat. II. Thymidine-radiographic study of the time of origin of neurons of the upper medulla, excluding the vestibular and auditory nuclei. J Comp Neurol. 1980a;194:37–56. - PubMed
-
- Altman J, Bayer SA. Development of the brain stem in the rat. IV. Thymidine-radiographic study of the time of origin of neurons in the pontine region. J Comp Neurol. 1980b;194:905–929. - PubMed
-
- Aoyama M, Hongo T, Kudo N, Tanaka R. Convergent effects from bilateral vestibulospinal tracts on spinal interneurons. Brain Res. 1971;35:250–253. - PubMed
-
- Auclair F, Bélanger MC, Marchand R. Ontogenetic study of early brain stem projections to the spinal cord in the rat. Brain Res Bull. 1993;30:281–289. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

