A dominant mutation in a neuronal acetylcholine receptor subunit leads to motor neuron degeneration in Caenorhabditis elegans
- PMID: 20962215
- PMCID: PMC2965043
- DOI: 10.1523/JNEUROSCI.1515-10.2010
A dominant mutation in a neuronal acetylcholine receptor subunit leads to motor neuron degeneration in Caenorhabditis elegans
Abstract
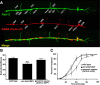
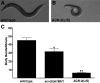
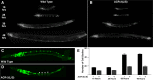
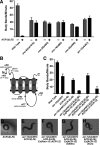
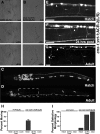
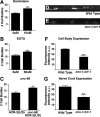
Inappropriate or excessive activation of ionotropic receptors can have dramatic consequences for neuronal function and, in many instances, leads to cell death. In Caenorhabditis elegans, nicotinic acetylcholine receptor (nAChR) subunits are highly expressed in a neural circuit that controls movement. Here, we show that heteromeric nAChRs containing the acr-2 subunit are diffusely localized in the processes of excitatory motor neurons and act to modulate motor neuron activity. Excessive signaling through these receptors leads to cell-autonomous degeneration of cholinergic motor neurons and paralysis. C. elegans double mutants lacking calreticulin and calnexin-two genes previously implicated in the cellular events leading to necrotic-like cell death (Xu et al. 2001)-are resistant to nAChR-mediated toxicity and possess normal numbers of motor neuron cell bodies. Nonetheless, excess nAChR activation leads to progressive destabilization of the motor neuron processes and, ultimately, paralysis in these animals. Our results provide new evidence that chronic activation of ionotropic receptors can have devastating degenerative effects in neurons and reveal that ion channel-mediated toxicity may have distinct consequences in neuronal cell bodies and processes.
Figures


References
-
- Brockie PJ, Mellem JE, Hills T, Madsen DM, Maricq AV. The C. elegans glutamate receptor subunit NMR-1 is required for slow NMDA-activated currents that regulate reversal frequency during locomotion. Neuron. 2001;31:617–630. - PubMed
-
- Chalfie M, Tu Y, Euskirchen G, Ward WW, Prasher DC. Green fluorescent protein as a marker for gene expression. Science. 1994;263:802–805. - PubMed
-
- Chase DL, Pepper JS, Koelle MR. Mechanism of extrasynaptic dopamine signaling in Caenorhabditis elegans. Nat Neurosci. 2004;7:1096–1103. - PubMed
-
- Cinar H, Keles S, Jin Y. Expression profiling of GABAergic motor neurons in Caenorhabditis elegans. Curr Biol. 2005;15:340–346. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
