Fibronectin-mediated upregulation of α5β1 integrin and cell adhesion during differentiation of mouse embryonic stem cells
- PMID: 20962574
- PMCID: PMC3038102
- DOI: 10.4161/cam.5.1.13704
Fibronectin-mediated upregulation of α5β1 integrin and cell adhesion during differentiation of mouse embryonic stem cells
Abstract
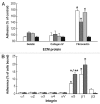
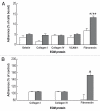
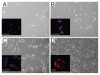
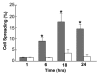
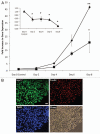
Embryonic stem (ES) cells have a broad potential application in regenerative medicine and can be differentiated into cells of all three germ layers. Adhesion of ES cells to extracellular matrix (ECM) proteins is essential for the differentiation pathway; Cell-ECM adhesion is mediated by integrins that have the ability to activate many intracellular signaling pathways. Therefore, we hypothesize that the expression and function of integrin receptors is a critical step in ES differentiation. Using functional cell adhesion assays, our study demonstrates that α5β1 is a major functional integrin receptor expressed on the cell surface of undifferentiated mouse ES-D3 cells, which showed significantly higher binding to fibronectin as compared to collagens. This adhesion was specific mediated by integrin α5β1 as evident from the inhibition with a disintegrin selective for this particular integrin. Differentiation of ES-D3 cells on fibronectin or on a collagen type1/fibronectin matrix, caused further selective up-regulation of the α5β1 integrin. Differentiation of the cells, as evaluated by immunofluorescence, FACS analysis and quantitative RT-PCR, was accompanied by the upregulation of mesenchymal (Flk1, isolectin B4, α-SMA, vimentin) and endodermal markers (FoxA2, SOX 17, cytokeratin) in parallel to increased expression of α5β1 integrin. Taken together, the data indicate that fibronectin-mediated, upregulation of α5β1 integrin and adhesion of ES-D3 cells to specific ECM molecules are linked to early stages of mouse embryonic stem cells commitment to meso-endodermal differentiation.
Figures

Similar articles
-
Deletion of mouse embryo fibroblast N-acetylglucosaminyltransferase V stimulates alpha5beta1 integrin expression mediated by the protein kinase C signaling pathway.J Biol Chem. 2005 Mar 4;280(9):8332-42. doi: 10.1074/jbc.M413532200. Epub 2004 Dec 22. J Biol Chem. 2005. PMID: 15615721
-
Alpha5beta1 integrin mediates strong tissue cohesion.J Cell Sci. 2003 Jan 15;116(Pt 2):377-86. doi: 10.1242/jcs.00231. J Cell Sci. 2003. PMID: 12482923
-
Regulation of ionizing radiation-induced adhesion of breast cancer cells to fibronectin by alpha5beta1 integrin.Radiat Res. 2014 Jun;181(6):650-8. doi: 10.1667/RR13543.1. Epub 2014 May 1. Radiat Res. 2014. PMID: 24785587 Free PMC article.
-
Fibronectin fibrillogenesis, a cell-mediated matrix assembly process.Matrix Biol. 2005 Sep;24(6):389-99. doi: 10.1016/j.matbio.2005.06.008. Matrix Biol. 2005. PMID: 16061370 Review.
-
Integrins in cell adhesion and signaling.Hum Cell. 1996 Sep;9(3):181-6. Hum Cell. 1996. PMID: 9183647 Review.
Cited by
-
Microenvironment influences vascular differentiation of murine cardiovascular progenitor cells.J Biomed Mater Res B Appl Biomater. 2014 Nov;102(8):1730-9. doi: 10.1002/jbm.b.33155. Epub 2014 Mar 31. J Biomed Mater Res B Appl Biomater. 2014. PMID: 24687591 Free PMC article.
-
Down regulation of ITGA4 and ITGA5 genes after formation of 3D spherules by human Wharton's jelly stem cells (hWJSCs).Mol Biol Rep. 2018 Jun;45(3):245-252. doi: 10.1007/s11033-018-4157-0. Epub 2018 Feb 6. Mol Biol Rep. 2018. PMID: 29411210
-
Efficient Isolation and Enrichment of Mesenchymal Stem Cells from Human Embryonic Stem Cells by Utilizing the Interaction between Integrin α5β1 and Fibronectin.Adv Sci (Weinh). 2020 Jul 19;7(17):2001365. doi: 10.1002/advs.202001365. eCollection 2020 Sep. Adv Sci (Weinh). 2020. PMID: 32995130 Free PMC article.
-
Hypoxia enhances differentiation of mouse embryonic stem cells into definitive endoderm and distal lung cells.Stem Cells Dev. 2015 Mar 1;24(5):663-76. doi: 10.1089/scd.2014.0343. Epub 2014 Oct 27. Stem Cells Dev. 2015. PMID: 25226206 Free PMC article.
-
Mechanotransduction through adhesion molecules: Emerging roles in regulating the stem cell niche.Front Cell Dev Biol. 2022 Sep 12;10:966662. doi: 10.3389/fcell.2022.966662. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 36172276 Free PMC article. Review.
References
-
- Keller G. Embryonic stem cell differentiation: Emergence of a new era in biology and medicine. Genes Dev. 2005;19:1129–1155. - PubMed
-
- Burdon T, Chambers I, Stracey C, Niwa H, Smith A. Signaling mechanisms regulating self-renewal and differentiation of pluripotent embryonic stem cells. Cells Tissues Organs. 1999;165:131–143. - PubMed
-
- Doyle B, Caplice N. A new source of endothelial progenitor cells—vascular biology redefined? Trends Biotechnol. 2005;23:444–446. - PubMed
-
- Doetschman TC, Eistetter H, Katz M, Schmidt W, Kemler R. The in vitro development of blastocyst-derived embryonic stem cell lines: Formation of visceral yolk sac, blood islands and myocardium. J Embryol Exp Morphol. 1985;87:27–45. - PubMed
-
- Toumadje A, Kusumoto K, Parton A, Mericko P, Dowell L, Ma G, et al. Pluripotent differentiation in vitro of murine ES-D3 embryonic stem cells. In Vitro Cell Dev Biol Anim. 2003;39:449–453. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
