Regulation of the mTOR complex 1 pathway by nutrients, growth factors, and stress
- PMID: 20965424
- PMCID: PMC2993060
- DOI: 10.1016/j.molcel.2010.09.026
Regulation of the mTOR complex 1 pathway by nutrients, growth factors, and stress
Abstract
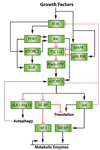
The large serine/threonine protein kinase mTOR regulates cellular and organismal homeostasis by coordinating anabolic and catabolic processes with nutrient, energy, and oxygen availability and growth factor signaling. Cells and organisms experience a wide variety of insults that perturb the homeostatic systems governed by mTOR and therefore require appropriate stress responses to allow cells to continue to function. Stress can manifest from an excess or lack of upstream signals or as a result of genetic perturbations in upstream effectors of the pathway. mTOR nucleates two large protein complexes that are important nodes in the pathways that help buffer cells from stresses, and are implicated in the progression of stress-associated phenotypes and diseases, such as aging, tumorigenesis, and diabetes. This review focuses on the key components of the mTOR complex 1 pathway and on how various stresses impinge upon them.
Copyright © 2010 Elsevier Inc. All rights reserved.
Figures

References
-
- Bailey CJ, Turner RC. Metformin. N Engl J Med. 1996;334:574–579. - PubMed
-
- Bentzinger CF, Romanino K, Cloetta D, Lin S, Mascarenhas JB, Oliveri F, Xia J, Casanova E, Costa CF, Brink M, et al. Skeletal muscle-specific ablation of raptor, but not of rictor, causes metabolic changes and results in muscle dystrophy. Cell Metab. 2008;8:411–424. - PubMed
-
- Bernardi R, Guernah I, Jin D, Grisendi S, Alimonti A, Teruya-Feldstein J, Cordon-Cardo C, Simon MC, Rafii S, Pandolfi PP. PML inhibits HIF-1alpha translation and neoangiogenesis through repression of mTOR. Nature. 2006;442:779–785. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

