Structure-based insights into the catalytic power and conformational dexterity of peroxiredoxins
- PMID: 20969484
- PMCID: PMC3125576
- DOI: 10.1089/ars.2010.3624
Structure-based insights into the catalytic power and conformational dexterity of peroxiredoxins
Abstract
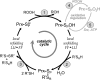
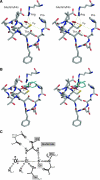
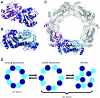
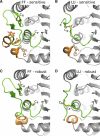
Peroxiredoxins (Prxs), some of nature's dominant peroxidases, use a conserved Cys residue to reduce peroxides. They are highly expressed in organisms from all kingdoms, and in eukaryotes they participate in hydrogen peroxide signaling. Seventy-two Prx structures have been determined that cover much of the diversity of the family. We review here the current knowledge and show that Prxs can be effectively classified by a structural/evolutionary organization into six subfamilies followed by specification of a 1-Cys or 2-Cys mechanism, and for 2-Cys Prxs, the structural location of the resolving Cys. We visualize the varied catalytic structural transitions and highlight how they differ depending on the location of the resolving Cys. We also review new insights into the question of how Prxs are such effective catalysts: the enzyme activates not only the conserved Cys thiolate but also the peroxide substrate. Moreover, the hydrogen-bonding network created by the four residues conserved in all Prx active sites stabilizes the transition state of the peroxidatic S(N)2 displacement reaction. Strict conservation of the peroxidatic active site along with the variation in structural transitions provides a fascinating picture of how the diverse Prxs function to break down peroxide substrates rapidly.
Figures
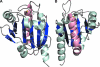

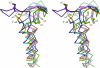
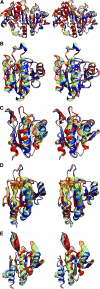
References
-
- Alphey MS. Bond CS. Tetaud E. Fairlamb AH. Hunter WN. The structure of reduced tryparedoxin peroxidase reveals a decamer and insight into reactivity of 2-Cys-peroxiredoxins. J Mol Biol. 2000;300:903–916. - PubMed
-
- Aran M. Ferrero DS. Pagano E. Wolosiuk RA. Typical 2-Cys peroxiredoxins: modulation by covalent transformations and noncovalent interactions. FEBS J. 2009;276:2478–2493. - PubMed
-
- Barranco-Medina S. Lazaro JJ. Dietz KJ. The oligomeric conformation of peroxiredoxins links redox state to function. FEBS Lett. 2009;583:1809–1816. - PubMed
-
- Biteau B. Labarre J. Toledano MB. ATP-dependent reduction of cysteine-sulphinic acid by S. cerevisiae sulphiredoxin. Nature. 2003;425:980–984. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

