Structural insight into the differential effects of omega-3 and omega-6 fatty acids on the production of Abeta peptides and amyloid plaques
- PMID: 20971855
- PMCID: PMC3057860
- DOI: 10.1074/jbc.M110.183608
Structural insight into the differential effects of omega-3 and omega-6 fatty acids on the production of Abeta peptides and amyloid plaques
Abstract
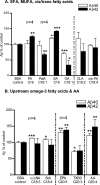
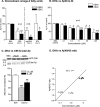
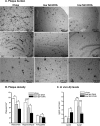
Several studies have shown the protective effects of dietary enrichment of various lipids in several late-onset animal models of Alzheimer Disease (AD); however, none of the studies has determined which structure within a lipid determines its detrimental or beneficial effects on AD. High-sensitivity enzyme-linked immunosorbent assay (ELISA) shows that saturated fatty acids (SFAs), upstream omega-3 FAs, and arachidonic acid (AA) resulted in significantly higher secretion of both Aβ 40 and 42 peptides compared with long chain downstream omega-3 and monounsaturated FAs (MUFA). Their distinct detrimental action is believed to be due to a structural template found in their fatty acyl chains that lack SFAs, upstream omega-3 FAs, and AA. Immunoblotting experiments and use of APP-C99-transfected COS-7 cells suggest that FA-driven altered production of Aβ is mediated through γ-secretase cleavage of APP. An early-onset AD transgenic mouse model expressing the double-mutant form of human amyloid precursor protein (APP); Swedish (K670N/M671L) and Indiana (V717F), corroborated in vitro findings by showing lower levels of Aβ and amyloid plaques in the brain, when they were fed a low fat diet enriched in DHA. Our work contributes to the clarification of aspects of structure-activity relationships.
Figures
References
-
- McLean C. A., Cherny R. A., Fraser F. W., Fuller S. J., Smith M. J., Beyreuther K., Bush A. I., Masters C. L. (1999) Ann. Neurol. 46, 860–866 - PubMed
-
- St George-Hyslop P. H. (2000) Sci. Am. 283, 76–83 - PubMed
-
- Grimm M. O., Grimm H. S., Pätzold A. J., Zinser E. G., Halonen R., Duering M., Tschäpe J. A., De Strooper B., Müller U., Shen J., Hartmann T. (2005) Nat. Cell Biol. 7, 1118–1123 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

