Genetic deletion of Nrf2 promotes immortalization and decreases life span of murine embryonic fibroblasts
- PMID: 20974733
- PMCID: PMC4007826
- DOI: 10.1093/gerona/glq181
Genetic deletion of Nrf2 promotes immortalization and decreases life span of murine embryonic fibroblasts
Abstract
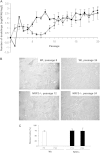
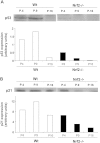
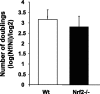
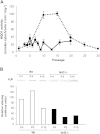
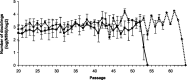
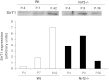
Nuclear factor E2-related factor-2 (Nrf2) transcription factor is one of the main regulators of intracellular redox balance and a sensor of oxidative and electrophilic stress. Low Nrf2 activity is usually associated with carcinogenesis, but Nrf2 is also considered as an oncogene because it increases survival of transformed cells. Because intracellular redox balance alterations are involved in both senescence and tumorigenesis, we investigated the impact of Nrf2 genetic deletion on cellular immortalization and life span of murine embryonic fibroblasts. We report that Nrf2 genetic deletion promotes immortalization due to an early loss of p53-dependent gene expression. However, compared with control cells, immortalized Nrf2-/- murine embryonic fibroblasts exhibited decreased growth, lower cyclin E levels, and impaired expression of NQO1 and cytochrome b₅ reductase. Moreover, SirT1 was also significantly reduced in immortalized Nrf2-/- murine embryonic fibroblasts, and these cells exhibited shorter life span. Our results underscore the dual role of Nrf2 in protection against carcinogenesis and in the delay of cellular aging.
Figures

References
-
- Hayflick L. The limited in vitro lifetime of human diploid cell strains. Exp Cell Res. 1965;37:614–636. - PubMed
-
- Sherr CJ, DePinho RA. Cellular senescence: mitotic clock or culture shock? Cell. 2000;102:407–410. - PubMed
-
- Dirac AM, Bernards R. Reversal of senescence in mouse fibroblasts through lentiviral suppression of p53. J Biol Chem. 2003;278:11731–11734. - PubMed
-
- Kiyono T. Molecular mechanisms of cellular senescence and immortalization of human cells. Expert Opin Ther Targets. 2007;11:1623–1637. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

