The lipid A phosphate position determines differential host Toll-like receptor 4 responses to phylogenetically related symbiotic and pathogenic bacteria
- PMID: 20974832
- PMCID: PMC3019871
- DOI: 10.1128/IAI.00937-10
The lipid A phosphate position determines differential host Toll-like receptor 4 responses to phylogenetically related symbiotic and pathogenic bacteria
Abstract
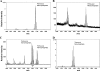
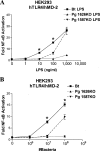
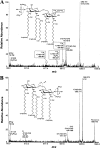
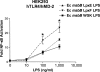
The human symbiont Bacteroides thetaiotaomicron promotes intestinal function and health, whereas the phylogenetically related pathogen Porphyromonas gingivalis is associated with the chronic oral inflammatory disease periodontitis. Although both B. thetaiotaomicron and P. gingivalis synthesize lipopolysaccharides (LPS) consisting of penta-acylated, monophosphorylated lipid A in addition to immunologically silent, nonphosphorylated lipid A, they elicit strikingly distinct Toll-like receptor 4 (TLR4) responses. We show that the phosphate position of penta-acylated, monophosphorylated lipid A is a key feature for determining the differential TLR4 responses elicited by these evolutionarily related bacteria. B. thetaiotaomicron produces TLR4-stimulatory lipid A bearing a 1-phosphate, in contrast to P. gingivalis, which produces TLR4-evasive lipid A bearing a 4'-phosphate. Confirming these observations, recombinant Escherichia coli LPS containing penta-acylated, 1-phosphorylated lipid A is more TLR4 stimulatory than LPS containing 4'-phosphorylated lipid A. The specific capacity of a Gram-negative bacterium to alert or evade the host innate immune defense system through TLR4-dependent signaling is currently recognized as a critical aspect defining the relationship between the host and the bacterium. We propose that the distinct lipid A phosphate positions observed for the B. thetaiotaomicron and P. gingivalis LPS contributes to the manifestation of these bacteria as commensal or pathogen within the human host.
Figures


References
-
- Bainbridge, B. W., S. R. Coats, T. T. Pham, R. A. Reife, and R. P. Darveau. 2006. Expression of a Porphyromonas gingivalis lipid A palmitylacyltransferase in Escherichia coli yields a chimeric lipid A with altered ability to stimulate interleukin-8 secretion. Cell Microbiol. 8:120-129. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

