Routine HIV screening in France: clinical impact and cost-effectiveness
- PMID: 20976112
- PMCID: PMC2956760
- DOI: 10.1371/journal.pone.0013132
Routine HIV screening in France: clinical impact and cost-effectiveness
Abstract
Background: In France, roughly 40,000 HIV-infected persons are unaware of their HIV infection. Although previous studies have evaluated the cost-effectiveness of routine HIV screening in the United States, differences in both the epidemiology of infection and HIV testing behaviors warrant a setting-specific analysis for France.
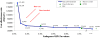
Methods/principal findings: We estimated the life expectancy (LE), cost and cost-effectiveness of alternative HIV screening strategies in the French general population and high-risk sub-populations using a computer model of HIV detection and treatment, coupled with French national clinical and economic data. We compared risk-factor-based HIV testing ("current practice") to universal routine, voluntary HIV screening in adults aged 18-69. Screening frequencies ranged from once to annually. Input data included mean age (42 years), undiagnosed HIV prevalence (0.10%), annual HIV incidence (0.01%), test acceptance (79%), linkage to care (75%) and cost/test (€43). We performed sensitivity analyses on HIV prevalence and incidence, cost estimates, and the transmission benefits of ART. "Current practice" produced LEs of 242.82 quality-adjusted life months (QALM) among HIV-infected persons and 268.77 QALM in the general population. Adding a one-time HIV screen increased LE by 0.01 QALM in the general population and increased costs by €50/person, for a cost-effectiveness ratio (CER) of €57,400 per quality-adjusted life year (QALY). More frequent screening in the general population increased survival, costs and CERs. Among injection drug users (prevalence 6.17%; incidence 0.17%/year) and in French Guyana (prevalence 0.41%; incidence 0.35%/year), annual screening compared to every five years produced CERs of €51,200 and €46,500/QALY.
Conclusions/significance: One-time routine HIV screening in France improves survival compared to "current practice" and compares favorably to other screening interventions recommended in Western Europe. In higher-risk groups, more frequent screening is economically justifiable.
Conflict of interest statement
Figures

References
-
- Cazein F, Pillonel J, Imounga L, Le Strat Y, Bousquet V, et al. Surveillance du dépistage et du diagnostic de l'infection VIH et du sida, France, 2008. Bulletin Epidemiologique Hebdomadaire Web. 2009;2
-
- Institut de Veille Sanitaire. HIV and AIDS surveillance in France, 2006. Special issue - HIV and AIDS infection in France and in Europe. Bulletin Epidemiologique Hebdomadaire. 2007:46–47: 386-393.
-
- Richet-Mastain L. 2006. Institut national de la statistique et des études économiques: Bilan démographique 2006: un excédent naturel record. Available: http://www.insee.fr/fr/ffc/ipweb/ip1118/ip1118.pdf. Accessed 14 December 2009.
-
- Conseil national du sida. Rapport sur l'évolution du dispositif de dépistage de l'infection par le VIH en France. 2006;14 Available: http://www.cns.sante.fr/htm/avis/rapports_pdf/06_11_16_depistage.pdf. Accessed 2009 May.
-
- Lanoy E, Mary-Krause M, Tattevin P, Perbost I, Poizot-Martin I, et al. Frequency, determinants and consequences of delayed access to care for HIV infection in France. Antivir Ther. 2007;12:89–96. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

