Biosynthesis and role of N-linked glycosylation in cell surface structures of archaea with a focus on flagella and s layers
- PMID: 20976295
- PMCID: PMC2952790
- DOI: 10.1155/2010/470138
Biosynthesis and role of N-linked glycosylation in cell surface structures of archaea with a focus on flagella and s layers
Abstract
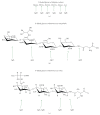
The genetics and biochemistry of the N-linked glycosylation system of Archaea have been investigated over the past 5 years using flagellins and S layers as reporter proteins in the model organisms, Methanococcus voltae, Methanococcus maripaludis, and Haloferax volcanii. Structures of archaeal N-linked glycans have indicated a variety of linking sugars as well as unique sugar components. In M. voltae, M. maripaludis, and H. volcanii, a number of archaeal glycosylation genes (agl) have been identified by deletion and complementation studies. These include many of the glycosyltransferases and the oligosaccharyltransferase needed to assemble the glycans as well as some of the genes encoding enzymes required for the biosynthesis of the sugars themselves. The N-linked glycosylation system is not essential for any of M. voltae, M. maripaludis, or H. volcanii, as demonstrated by the successful isolation of mutants carrying deletions in the oligosaccharyltransferase gene aglB (a homologue of the eukaryotic Stt3 subunit of the oligosaccharyltransferase complex). However, mutations that affect the glycan structure have serious effects on both flagellation and S layer function.
Figures



Similar articles
-
Complementation of an aglB Mutant of Methanococcus maripaludis with Heterologous Oligosaccharyltransferases.PLoS One. 2016 Dec 1;11(12):e0167611. doi: 10.1371/journal.pone.0167611. eCollection 2016. PLoS One. 2016. PMID: 27907170 Free PMC article.
-
N-glycosylation of Haloferax volcanii flagellins requires known Agl proteins and is essential for biosynthesis of stable flagella.J Bacteriol. 2012 Sep;194(18):4876-87. doi: 10.1128/JB.00731-12. Epub 2012 Jun 22. J Bacteriol. 2012. PMID: 22730124 Free PMC article.
-
Identification of genes involved in the biosynthesis and attachment of Methanococcus voltae N-linked glycans: insight into N-linked glycosylation pathways in Archaea.Mol Microbiol. 2006 Jul;61(1):259-68. doi: 10.1111/j.1365-2958.2006.05226.x. Mol Microbiol. 2006. PMID: 16824110
-
S-layer glycoproteins and flagellins: reporters of archaeal posttranslational modifications.Archaea. 2010 Jul 20;2010:612948. doi: 10.1155/2010/612948. Archaea. 2010. PMID: 20721273 Free PMC article. Review.
-
Hot and sweet: protein glycosylation in Crenarchaeota.Biochem Soc Trans. 2013 Feb 1;41(1):384-92. doi: 10.1042/BST20120296. Biochem Soc Trans. 2013. PMID: 23356316 Review.
Cited by
-
N-linked glycosylation in Archaea: a structural, functional, and genetic analysis.Microbiol Mol Biol Rev. 2014 Jun;78(2):304-41. doi: 10.1128/MMBR.00052-13. Microbiol Mol Biol Rev. 2014. PMID: 24847024 Free PMC article. Review.
-
Analysis of the cell surface layer ultrastructure of the oral pathogen Tannerella forsythia.Arch Microbiol. 2012 Jun;194(6):525-39. doi: 10.1007/s00203-012-0792-3. Epub 2012 Jan 25. Arch Microbiol. 2012. PMID: 22273979 Free PMC article.
-
Culture- and metagenomics-enabled analyses of the Methanosphaera genus reveals their monophyletic origin and differentiation according to genome size.ISME J. 2018 Dec;12(12):2942-2953. doi: 10.1038/s41396-018-0225-7. Epub 2018 Aug 1. ISME J. 2018. PMID: 30068938 Free PMC article.
-
Archaeosortases and exosortases are widely distributed systems linking membrane transit with posttranslational modification.J Bacteriol. 2012 Jan;194(1):36-48. doi: 10.1128/JB.06026-11. Epub 2011 Oct 28. J Bacteriol. 2012. PMID: 22037399 Free PMC article.
-
Complementation of an aglB Mutant of Methanococcus maripaludis with Heterologous Oligosaccharyltransferases.PLoS One. 2016 Dec 1;11(12):e0167611. doi: 10.1371/journal.pone.0167611. eCollection 2016. PLoS One. 2016. PMID: 27907170 Free PMC article.
References
-
- Szymanski CM, Wren BW. Protein glycosylation in bacterial mucosal pathogens. Nature Reviews Microbiology. 2005;3(3):225–237. - PubMed
-
- Mescher MF, Strominger JL. Purification and characterization of a prokaryotic glycoprotein from the cell envelope of Halobacterium salinarium . Journal of Biological Chemistry. 1976;251(7):2005–2014. - PubMed
-
- Magidovich H, Eichler J. Glycosyltransferases and oligosaccharyltransferases in Archaea: putative components of the N-glycosylation pathway in the third domain of life. FEMS Microbiology Letters. 2009;300(1):122–130. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

