Heparanase enhances local and systemic osteolysis in multiple myeloma by upregulating the expression and secretion of RANKL
- PMID: 20978204
- PMCID: PMC2970667
- DOI: 10.1158/0008-5472.CAN-10-2179
Heparanase enhances local and systemic osteolysis in multiple myeloma by upregulating the expression and secretion of RANKL
Abstract
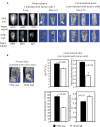
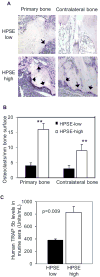
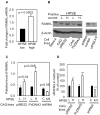
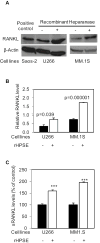
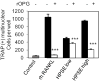
Excessive bone destruction is a major cause of morbidity in myeloma patients. However, the biological mechanisms involved in the pathogenesis of myeloma-induced bone disease are not fully understood. Heparanase, an enzyme that cleaves the heparan sulfate chains of proteoglycans, is upregulated in a variety of human tumors, including multiple myeloma. We previously showed that heparanase promotes robust myeloma tumor growth and supports spontaneous metastasis of tumor cells to bone. In the present study, we show, for the first time, that the expression of heparanase by myeloma tumor cells remarkably enhances bone destruction locally within the tumor microenvironment. In addition, enhanced heparanase expression in the primary tumor also stimulated systemic osteoclastogenesis and osteolysis, thus mimicking the systemic osteoporosis often seen in myeloma patients. These effects occur, at least in part, as the result of a significant elevation in the expression and secretion of receptor activator of NF-κB ligand (RANKL) by heparanase-expressing myeloma cells. Moreover, analysis of bone marrow biopsies from myeloma patients reveals a positive correlation between the level of expression of heparanase and RANKL. Together, these discoveries reveal a novel and key role for heparanase in promoting tumor osteolysis and show that RANKL is central to the mechanism of heparanase-mediated osteolysis in myeloma.
©2010 AACR.
Conflict of interest statement
Figures


Similar articles
-
High heparanase activity in multiple myeloma is associated with elevated microvessel density.Cancer Res. 2003 Dec 15;63(24):8749-56. Cancer Res. 2003. PMID: 14695190
-
Heparanase inhibits osteoblastogenesis and shifts bone marrow progenitor cell fate in myeloma bone disease.Bone. 2013 Nov;57(1):10-7. doi: 10.1016/j.bone.2013.07.024. Epub 2013 Jul 27. Bone. 2013. PMID: 23895995 Free PMC article.
-
Heparan sulfate proteoglycans and heparanase--partners in osteolytic tumor growth and metastasis.Matrix Biol. 2004 Oct;23(6):341-52. doi: 10.1016/j.matbio.2004.08.004. Matrix Biol. 2004. PMID: 15533755 Review.
-
Critical role of AKT protein in myeloma-induced osteoclast formation and osteolysis.J Biol Chem. 2013 Oct 18;288(42):30399-30410. doi: 10.1074/jbc.M113.469973. Epub 2013 Sep 4. J Biol Chem. 2013. PMID: 24005670 Free PMC article. Clinical Trial.
-
New insights in myeloma-induced osteolysis.Leuk Lymphoma. 2003 Sep;44(9):1463-7. doi: 10.3109/10428190309178765. Leuk Lymphoma. 2003. PMID: 14565645 Review.
Cited by
-
Myeloma cell-derived Runx2 promotes myeloma progression in bone.Blood. 2015 Jun 4;125(23):3598-608. doi: 10.1182/blood-2014-12-613968. Epub 2015 Apr 10. Blood. 2015. PMID: 25862559 Free PMC article.
-
Key Matrix Remodeling Enzymes: Functions and Targeting in Cancer.Cancers (Basel). 2021 Mar 22;13(6):1441. doi: 10.3390/cancers13061441. Cancers (Basel). 2021. PMID: 33809973 Free PMC article. Review.
-
The potential of heparanase as a therapeutic target in cancer.Biochem Pharmacol. 2014 May 1;89(1):12-9. doi: 10.1016/j.bcp.2014.02.010. Epub 2014 Feb 22. Biochem Pharmacol. 2014. PMID: 24565907 Free PMC article.
-
Mesenchymal stem cells expressing osteoprotegerin variants inhibit osteolysis in a murine model of multiple myeloma.Blood Adv. 2017 Nov 21;1(25):2375-2385. doi: 10.1182/bloodadvances.2017007310. eCollection 2017 Nov 28. Blood Adv. 2017. PMID: 29296887 Free PMC article.
-
Animal Models of Bone Metastasis.Vet Pathol. 2015 Sep;52(5):827-41. doi: 10.1177/0300985815586223. Epub 2015 May 28. Vet Pathol. 2015. PMID: 26021553 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

