Neuronal MCP-1 mediates microglia recruitment and neurodegeneration induced by the mild impairment of oxidative metabolism
- PMID: 21029241
- PMCID: PMC3046243
- DOI: 10.1111/j.1750-3639.2010.00445.x
Neuronal MCP-1 mediates microglia recruitment and neurodegeneration induced by the mild impairment of oxidative metabolism
Abstract
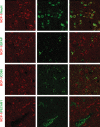
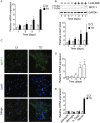
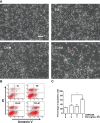
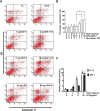
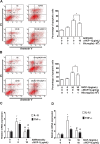
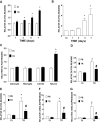
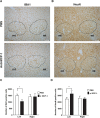
Chemokines are implicated in the neuroinflammation of several chronic neurodegenerative disorders. However, the precise role of chemokines in neurodegeneration is unknown. Thiamine deficiency (TD) causes abnormal oxidative metabolism in the brain as well as a well-defined microglia activation and neurodegeneration in the submedial thalamus nucleus (SmTN), which are common features of neurodegenerative diseases. We evaluated the role of chemokines in neurodegeneration and the underlying mechanism in a TD model. Among the chemokines examined, TD selectively induced neuronal expression of monocyte chemoattractant protein-1 (MCP-1) in the SmTN prior to microglia activation and neurodegeneration. The conditioned medium collected from TD-induced neurons caused microglia activation. With a neuron/microglia co-culture system, we showed that MCP-1-induced neurotoxicity required the presence of microglia, and exogenous MCP-1 was able to activate microglia and stimulated microglia to produce cytokines. A MCP-1 neutralizing antibody inhibited MCP-1-induced microglia activation and neuronal death in culture and in the thalamus. MCP-1 knockout mice were resistant to TD-induced neuronal death in SmTN. TD selectively induced the accumulation of reactive oxygen species in neurons, and antioxidants blocked TD-induced MCP-1 expression. Together, our results indicated an induction of neuronal MCP-1 during mild impairment of oxidative metabolism caused by microglia recruitment/activation, which exacerbated neurodegeneration.
© 2010 The Authors; Brain Pathology © 2010 International Society of Neuropathology.
Figures




Similar articles
-
Translocation of amyloid precursor protein C-terminal fragment(s) to the nucleus precedes neuronal death due to thiamine deficiency-induced mild impairment of oxidative metabolism.Neurochem Res. 2008 Jul;33(7):1365-72. doi: 10.1007/s11064-008-9594-z. Epub 2008 Mar 4. Neurochem Res. 2008. PMID: 18317926 Free PMC article.
-
Autophagy alleviates neurodegeneration caused by mild impairment of oxidative metabolism.J Neurochem. 2013 Sep;126(6):805-18. doi: 10.1111/jnc.12268. Epub 2013 Jun 9. J Neurochem. 2013. PMID: 23586593 Free PMC article.
-
CD40-CD40L interactions promote neuronal death in a model of neurodegeneration due to mild impairment of oxidative metabolism.Neurochem Int. 2005 Aug;47(3):204-15. doi: 10.1016/j.neuint.2005.03.002. Neurochem Int. 2005. PMID: 15885854
-
Selective response of various brain cell types during neurodegeneration induced by mild impairment of oxidative metabolism.Neurochem Int. 2004 Jul-Aug;45(2-3):361-9. doi: 10.1016/j.neuint.2003.09.008. Neurochem Int. 2004. PMID: 15145550 Review.
-
Role of MCP-1 and CCR2 in alcohol neurotoxicity.Pharmacol Res. 2019 Jan;139:360-366. doi: 10.1016/j.phrs.2018.11.030. Epub 2018 Nov 22. Pharmacol Res. 2019. PMID: 30472461 Free PMC article. Review.
Cited by
-
Change in psychiatric symptomatology after benfotiamine treatment in males is related to lifetime alcoholism severity.Drug Alcohol Depend. 2015 Jul 1;152:257-63. doi: 10.1016/j.drugalcdep.2015.03.032. Epub 2015 Apr 8. Drug Alcohol Depend. 2015. PMID: 25908323 Free PMC article. Clinical Trial.
-
From Organotypic Mouse Brain Slices to Human Alzheimer Plasma Biomarkers: A Focus on Microglia.Biomolecules. 2024 Sep 3;14(9):1109. doi: 10.3390/biom14091109. Biomolecules. 2024. PMID: 39334874 Free PMC article.
-
Toll-like receptor 4 stimulation with monophosphoryl lipid A ameliorates motor deficits and nigral neurodegeneration triggered by extraneuronal α-synucleinopathy.Mol Neurodegener. 2017 Jul 4;12(1):52. doi: 10.1186/s13024-017-0195-7. Mol Neurodegener. 2017. PMID: 28676095 Free PMC article.
-
WKYMVm/FPR2 Alleviates Spinal Cord Injury by Attenuating the Inflammatory Response of Microglia.Mediators Inflamm. 2022 Jul 27;2022:4408099. doi: 10.1155/2022/4408099. eCollection 2022. Mediators Inflamm. 2022. PMID: 35935810 Free PMC article.
-
Time-Dependent Protective and Pro-Resolving Effects of FPR2 Agonists on Lipopolysaccharide-Exposed Microglia Cells Involve Inhibition of NF-κB and MAPKs Pathways.Cells. 2021 Sep 9;10(9):2373. doi: 10.3390/cells10092373. Cells. 2021. PMID: 34572022 Free PMC article.
References
-
- Allan SM, Rothwell NJ (2001) Cytokines and acute neurodegeneration. Nat Rev Neurosci 2:734–744. - PubMed
-
- Antony JM, van Marle G, Opii W, Butterfield DA, Mallet F, Yong VW et al (2004) Human endogenous retrovirus glycoprotein—mediated induction of redox reactants causes oligodendrocyte death and demyelination. Nat Neurosci 7:1088–1095. - PubMed
-
- Banisadr G, Gosselin RD, Mechighel P, Kitabgi P, Rostene W, Parsadaniantz SM (2005) Highly regionalized neuronal expression of monocyte chemoattractant protein‐1 (MCP‐1/CCL2) in rat brain: evidence for its colocalization with neurotransmitters and neuropeptides. J Comp Neurol 489:275–292. - PubMed
-
- Benveniste EN, Nguyen VT, O'Keefe GM (2001) Immunological aspects of microglia: relevance to Alzheimer's disease. Neurochem Int 39:381–391. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

