Spiral ligament fibrocyte-derived MCP-1/CCL2 contributes to inner ear inflammation secondary to nontypeable H. influenzae-induced otitis media
- PMID: 21029462
- PMCID: PMC2988798
- DOI: 10.1186/1471-2334-10-314
Spiral ligament fibrocyte-derived MCP-1/CCL2 contributes to inner ear inflammation secondary to nontypeable H. influenzae-induced otitis media
Abstract
Background: Otitis media (OM), one of the most common pediatric infectious diseases, causes inner ear inflammation resulting in vertigo and sensorineural hearing loss. Previously, we showed that spiral ligament fibrocytes (SLFs) recognize OM pathogens and up-regulate chemokines. Here, we aim to determine a key molecule derived from SLFs, contributing to OM-induced inner ear inflammation.
Methods: Live NTHI was injected into the murine middle ear through the tympanic membrane, and histological analysis was performed after harvesting the temporal bones. Migration assays were conducted using the conditioned medium of NTHI-exposed SLFs with and without inhibition of MCP-1/CCL2 and CCR2. qRT-PCR analysis was performed to demonstrate a compensatory up-regulation of alternative genes induced by the targeting of MCP-1/CCL2 or CCR2.
Results: Transtympanic inoculation of live NTHI developed serous and purulent labyrinthitis after clearance of OM. THP-1 cells actively migrated and invaded the extracellular matrix in response to the conditioned medium of NTHI-exposed SLFs. This migratory activity was markedly inhibited by the viral CC chemokine inhibitor and the deficiency of MCP-1/CCL2, indicating that MCP-1/CCL2 is a main attractant of THP-1 cells among the SLF-derived molecules. We further demonstrated that CCR2 deficiency inhibits migration of monocyte-like cells in response to NTHI-induced SLF-derived molecules. Immunolabeling showed an increase in MCP-1/CCL2 expression in the cochlear lateral wall of the NTHI-inoculated group. Contrary to the in vitro data, deficiency of MCP-1/CCL2 or CCR2 did not inhibit OM-induced inner ear inflammation in vivo. We demonstrated that targeting MCP-1/CCL2 enhances NTHI-induced up-regulation of MCP-2/CCL8 in SLFs and up-regulates the basal expression of CCR2 in the splenocytes. We also found that targeting CCR2 enhances NTHI-induced up-regulation of MCP-1/CCL2 in SLFs.
Conclusions: Taken together, we suggest that NTHI-induced SLF-derived MCP-1/CCL2 is a key molecule contributing to inner ear inflammation through CCR2-mediated recruitment of monocytes. However, deficiency of MCP-1/CCL2 or CCR2 alone was limited to inhibit OM-induced inner ear inflammation due to compensation of alternative genes.
Figures

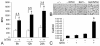
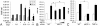

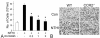
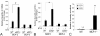
Similar articles
-
IL-10/HMOX1 signaling modulates cochlear inflammation via negative regulation of MCP-1/CCL2 expression in cochlear fibrocytes.J Immunol. 2015 Apr 15;194(8):3953-61. doi: 10.4049/jimmunol.1402751. Epub 2015 Mar 16. J Immunol. 2015. PMID: 25780042 Free PMC article.
-
Toll-like receptor 2-dependent NF-kappaB activation is involved in nontypeable Haemophilus influenzae-induced monocyte chemotactic protein 1 up-regulation in the spiral ligament fibrocytes of the inner ear.Infect Immun. 2007 Jul;75(7):3361-72. doi: 10.1128/IAI.01886-06. Epub 2007 Apr 23. Infect Immun. 2007. PMID: 17452470 Free PMC article.
-
Spiral ligament fibrocytes release chemokines in response to otitis media pathogens.Acta Otolaryngol. 2006 Jun;126(6):564-9. doi: 10.1080/00016480500452525. Acta Otolaryngol. 2006. PMID: 16720438
-
A CCL2-based fusokine as a novel biopharmaceutical for the treatment of CCR2-driven autoimmune diseases.Crit Rev Immunol. 2010;30(5):449-61. doi: 10.1615/critrevimmunol.v30.i5.40. Crit Rev Immunol. 2010. PMID: 21083526 Review.
-
The adhesins of non-typeable Haemophilus influenzae.Expert Rev Anti Infect Ther. 2018 Mar;16(3):187-196. doi: 10.1080/14787210.2018.1438263. Epub 2018 Feb 21. Expert Rev Anti Infect Ther. 2018. PMID: 29415569 Review.
Cited by
-
IL-10/HMOX1 signaling modulates cochlear inflammation via negative regulation of MCP-1/CCL2 expression in cochlear fibrocytes.J Immunol. 2015 Apr 15;194(8):3953-61. doi: 10.4049/jimmunol.1402751. Epub 2015 Mar 16. J Immunol. 2015. PMID: 25780042 Free PMC article.
-
NOD2/RICK-dependent β-defensin 2 regulation is protective for nontypeable Haemophilus influenzae-induced middle ear infection.PLoS One. 2014 Mar 13;9(3):e90933. doi: 10.1371/journal.pone.0090933. eCollection 2014. PLoS One. 2014. PMID: 24625812 Free PMC article.
-
Biology and pathobiology of lipid droplets and their potential role in the protection of the organ of Corti.Hear Res. 2015 Dec;330(Pt A):26-38. doi: 10.1016/j.heares.2015.04.015. Epub 2015 May 15. Hear Res. 2015. PMID: 25987503 Free PMC article. Review.
-
Autoinflammatory characteristics and short-term effects of delivering high-dose steroids to the surface of the intact endolymphatic sac and incus in refractory Ménière's disease.J Otol. 2019 Jun;14(2):40-50. doi: 10.1016/j.joto.2019.01.001. Epub 2019 Jan 6. J Otol. 2019. PMID: 31223300 Free PMC article.
-
Ultrastructure and three-dimensional architecture of the anterior cruciate ligament in the knee joints of young and old monkeys.Med Mol Morphol. 2020 Mar;53(1):7-14. doi: 10.1007/s00795-019-00224-7. Epub 2019 May 18. Med Mol Morphol. 2020. PMID: 31104131
References
-
- Paparella MM, Morizono T, Le CT, Mancini F, Sipila P, Choo YB, Liden G, Kim CS. Sensorineural hearing loss in otitis media. Ann Otol Rhinol Laryngol. 1984;93:623–629. - PubMed
-
- Casselbrant ML, Furman JM, Rubenstein E, Mandel EM. Effect of otitis media on the vestibular system in children. Ann Otol Rhinol Laryngol. 1995;104:620–624. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

