Regulating DnaA complex assembly: it is time to fill the gaps
- PMID: 21035377
- PMCID: PMC3005629
- DOI: 10.1016/j.mib.2010.10.001
Regulating DnaA complex assembly: it is time to fill the gaps
Abstract
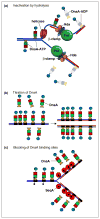
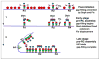
New rounds of bacterial chromosome replication are triggered during each cell division cycle by the initiator protein, DnaA. For precise timing, interactions of DnaA-ATP monomers with the replication origin, oriC, must be carefully regulated during formation of complexes that unwind origin DNA and load replicative helicase. Recent studies in Escherichia coli suggest that high and low affinity DnaA recognition sites are positioned within oriC to direct staged assembly of bacterial pre-replication complexes, with DnaA contacting low affinity sites as it oligomerizes to 'fill the gaps' between high affinity sites. The wide variability of oriC DnaA recognition site patterns seen in nature may reflect myriad gap-filling strategies needed to couple oriC function to the lifestyle of different bacterial types.
Copyright © 2010 Elsevier Ltd. All rights reserved.
Figures

Similar articles
-
Two oppositely oriented arrays of low-affinity recognition sites in oriC guide progressive binding of DnaA during Escherichia coli pre-RC assembly.Mol Microbiol. 2011 Oct;82(2):475-88. doi: 10.1111/j.1365-2958.2011.07827.x. Epub 2011 Sep 14. Mol Microbiol. 2011. PMID: 21895796 Free PMC article.
-
Building the bacterial orisome: high-affinity DnaA recognition plays a role in setting the conformation of oriC DNA.Mol Microbiol. 2014 Mar;91(6):1148-63. doi: 10.1111/mmi.12525. Epub 2014 Feb 18. Mol Microbiol. 2014. PMID: 24443848 Free PMC article.
-
SeqA blocking of DnaA-oriC interactions ensures staged assembly of the E. coli pre-RC.Mol Cell. 2006 Nov 17;24(4):581-92. doi: 10.1016/j.molcel.2006.09.016. Mol Cell. 2006. PMID: 17114060 Free PMC article.
-
Regulation of DnaA assembly and activity: taking directions from the genome.Annu Rev Microbiol. 2011;65:19-35. doi: 10.1146/annurev-micro-090110-102934. Annu Rev Microbiol. 2011. PMID: 21639790 Free PMC article. Review.
-
Regulating DNA replication in bacteria.Cold Spring Harb Perspect Biol. 2013 Apr 1;5(4):a012922. doi: 10.1101/cshperspect.a012922. Cold Spring Harb Perspect Biol. 2013. PMID: 23471435 Free PMC article. Review.
Cited by
-
Topological characterization of the DnaA-oriC complex using single-molecule nanomanipuation.Nucleic Acids Res. 2012 Aug;40(15):7375-83. doi: 10.1093/nar/gks371. Epub 2012 May 11. Nucleic Acids Res. 2012. PMID: 22581769 Free PMC article.
-
Interactions of Escherichia coli molecular chaperone HtpG with DnaA replication initiator DNA.Cell Stress Chaperones. 2015 Nov;20(6):951-7. doi: 10.1007/s12192-015-0623-y. Epub 2015 Aug 6. Cell Stress Chaperones. 2015. PMID: 26246199 Free PMC article.
-
The primosomal protein DnaD inhibits cooperative DNA binding by the replication initiator DnaA in Bacillus subtilis.J Bacteriol. 2012 Sep;194(18):5110-7. doi: 10.1128/JB.00958-12. Epub 2012 Jul 20. J Bacteriol. 2012. PMID: 22821970 Free PMC article.
-
Depletion of acidic phospholipids influences chromosomal replication in Escherichia coli.Microbiologyopen. 2012 Dec;1(4):450-66. doi: 10.1002/mbo3.46. Epub 2012 Nov 16. Microbiologyopen. 2012. PMID: 23233230 Free PMC article.
-
Cell Size Is Coordinated with Cell Cycle by Regulating Initiator Protein DnaA in E. coli.Biophys J. 2020 Dec 15;119(12):2537-2557. doi: 10.1016/j.bpj.2020.10.034. Epub 2020 Nov 13. Biophys J. 2020. PMID: 33189684 Free PMC article.
References
-
- Fuller RS, Funnell BE, Kornberg A. The dnaA protein complex with the E. coli chromosomal replication origin (oriC) and other DNA sites. Cell. 1984;38:889–900. - PubMed
-
- Crooke E, Thresher R, Hwang DS, Griffith J, Kornberg A. Replicatively active complexes of DnaA protein and the Escherichia coli chromosomal origin observed in the electron microscope. J Mol Biol. 1993;233:16–24. - PubMed
-
- Margulies C, Kaguni JM. Ordered and sequential binding of DnaA protein to oriC, the chromosomal origin of Escherichia coli. J Biol Chem. 1996;271:17035–17040. - PubMed
-
- Kawakami H, Katayama T. DnaA, ORC, and Cdc6: similarity beyond the domains of life and diversity. Biochem Cell Biol. 2010;88:49–62. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

