Calcium binding kinetics of troponin C strongly modulate cooperative activation and tension kinetics in cardiac muscle
- PMID: 21035455
- PMCID: PMC3018540
- DOI: 10.1016/j.yjmcc.2010.10.025
Calcium binding kinetics of troponin C strongly modulate cooperative activation and tension kinetics in cardiac muscle
Abstract
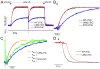
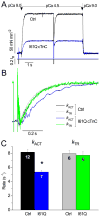
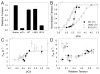
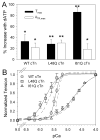
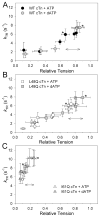
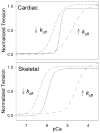
Tension development and relaxation in cardiac muscle are regulated at the thin filament via Ca(2+) binding to cardiac troponin C (cTnC) and strong cross-bridge binding. However, the influence of cTnC Ca(2+)-binding properties on these processes in the organized structure of cardiac sarcomeres is not well-understood and likely differs from skeletal muscle. To study this we generated single amino acid variants of cTnC with altered Ca(2+) dissociation rates (k(off)), as measured in whole troponin (cTn) complex by stopped-flow spectroscopy (I61Q cTn>WT cTn>L48Q cTn), and exchanged them into cardiac myofibrils and demembranated trabeculae. In myofibrils at saturating Ca(2+), L48Q cTnC did not affect maximum tension (T(max)), thin filament activation (k(ACT)) and tension development (k(TR)) rates, or the rates of relaxation, but increased duration of slow phase relaxation. In contrast, I61Q cTnC reduced T(max), k(ACT) and k(TR) by 40-65% with little change in relaxation. Interestingly, k(ACT) was less than k(TR) with I61Q cTnC, and this difference increased with addition of inorganic phosphate, suggesting that reduced cTnC Ca(2+)-affinity can limit thin filament activation kinetics. Trabeculae exchanged with I61Q cTn had reduced T(max), Ca(2+) sensitivity of tension (pCa(50)), and slope (n(H)) of tension-pCa, while L48Q cTn increased pCa(50) and reduced n(H). Increased cross-bridge cycling with 2-deoxy-ATP increased pCa(50) with WT or L48Q cTn, but not I61Q cTn. We discuss the implications of these results for understanding the role of cTn Ca(2+)-binding properties on the magnitude and rate of tension development and relaxation in cardiac muscle.
Copyright © 2010 Elsevier Ltd. All rights reserved.
Figures
Similar articles
-
Thin filament incorporation of an engineered cardiac troponin C variant (L48Q) enhances contractility in intact cardiomyocytes from healthy and infarcted hearts.J Mol Cell Cardiol. 2014 Jul;72:219-27. doi: 10.1016/j.yjmcc.2014.03.015. Epub 2014 Mar 29. J Mol Cell Cardiol. 2014. PMID: 24690333 Free PMC article.
-
Enhanced Ca2+ binding of cardiac troponin reduces sarcomere length dependence of contractile activation independently of strong crossbridges.Am J Physiol Heart Circ Physiol. 2012 Oct 1;303(7):H863-70. doi: 10.1152/ajpheart.00395.2012. Epub 2012 Aug 3. Am J Physiol Heart Circ Physiol. 2012. PMID: 22865385 Free PMC article.
-
The effect of variable troponin C mutation thin filament incorporation on cardiac muscle twitch contractions.J Mol Cell Cardiol. 2021 Jun;155:112-124. doi: 10.1016/j.yjmcc.2021.02.009. Epub 2021 Feb 24. J Mol Cell Cardiol. 2021. PMID: 33636222 Free PMC article.
-
Mouse Models of Cardiomyopathies Caused by Mutations in Troponin C.Int J Mol Sci. 2023 Aug 2;24(15):12349. doi: 10.3390/ijms241512349. Int J Mol Sci. 2023. PMID: 37569724 Free PMC article. Review.
-
Ca(2+) exchange with troponin C and cardiac muscle dynamics.Cardiovasc Res. 2008 Mar 1;77(4):619-26. doi: 10.1093/cvr/cvm098. Epub 2007 Dec 12. Cardiovasc Res. 2008. PMID: 18079104 Review.
Cited by
-
Stromal Cells in Dense Collagen Promote Cardiomyocyte and Microvascular Patterning in Engineered Human Heart Tissue.Tissue Eng Part A. 2016 Apr;22(7-8):633-44. doi: 10.1089/ten.TEA.2015.0482. Epub 2016 Mar 31. Tissue Eng Part A. 2016. PMID: 26955856 Free PMC article.
-
Structural determinants of muscle thin filament cooperativity.Arch Biochem Biophys. 2016 Mar 15;594:8-17. doi: 10.1016/j.abb.2016.02.016. Epub 2016 Feb 15. Arch Biochem Biophys. 2016. PMID: 26891592 Free PMC article. Review.
-
Length dependence of striated muscle force generation is controlled by phosphorylation of cTnI at serines 23/24.J Physiol. 2013 Sep 15;591(18):4535-47. doi: 10.1113/jphysiol.2013.258400. Epub 2013 Jul 8. J Physiol. 2013. PMID: 23836688 Free PMC article.
-
Thin filament incorporation of an engineered cardiac troponin C variant (L48Q) enhances contractility in intact cardiomyocytes from healthy and infarcted hearts.J Mol Cell Cardiol. 2014 Jul;72:219-27. doi: 10.1016/j.yjmcc.2014.03.015. Epub 2014 Mar 29. J Mol Cell Cardiol. 2014. PMID: 24690333 Free PMC article.
-
Computational studies of the effect of the S23D/S24D troponin I mutation on cardiac troponin structural dynamics.Biophys J. 2014 Oct 7;107(7):1675-85. doi: 10.1016/j.bpj.2014.08.008. Biophys J. 2014. PMID: 25296321 Free PMC article.
References
-
- Gordon AM, Homsher E, Regnier M. Regulation of contraction in striated muscle. Physiol Rev. 2000 Apr;80(2):853–924. - PubMed
-
- Hofmann PA, Fuchs F. Evidence for a force-dependent component of calcium binding to cardiac troponin C. Am J Physiol. 1987 Oct;253(4 Pt 1):C541–6. - PubMed
-
- Poggesi C, Tesi C, Stehle R. Sarcomeric determinants of striated muscle relaxation kinetics. Pflugers Arch. 2005 Mar;449(6):505–17. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

