Zinc transporter 3 is involved in learned fear and extinction, but not in innate fear
- PMID: 21036893
- PMCID: PMC2981414
- DOI: 10.1101/lm.1962010
Zinc transporter 3 is involved in learned fear and extinction, but not in innate fear
Abstract
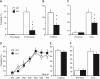
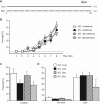
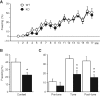
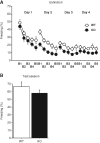
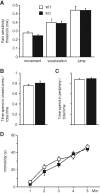
Synaptically released Zn²+ is a potential modulator of neurotransmission and synaptic plasticity in fear-conditioning pathways. Zinc transporter 3 (ZnT3) knock-out (KO) mice are well suited to test the role of zinc in learned fear, because ZnT3 is colocalized with synaptic zinc, responsible for its transport to synaptic vesicles, highly enriched in the amygdala-associated neural circuitry, and ZnT3 KO mice lack Zn²+ in synaptic vesicles. However, earlier work reported no deficiency in fear memory in ZnT3 KO mice, which is surprising based on the effects of Zn²+ on amygdala synaptic plasticity. We therefore reexamined ZnT3 KO mice in various tasks for learned and innate fear. The mutants were deficient in a weak fear-conditioning protocol using single tone-shock pairing but showed normal memory when a stronger, five-pairing protocol was used. ZnT3 KO mice were deficient in memory when a tone was presented as complex auditory information in a discontinuous fashion. Moreover, ZnT3 KO mice showed abnormality in trace fear conditioning and in fear extinction. By contrast, ZnT3 KO mice had normal anxiety. Thus, ZnT3 is involved in associative fear memory and extinction, but not in innate fear, consistent with the role of synaptic zinc in amygdala synaptic plasticity.
Figures

Similar articles
-
Behavioral characterization of female zinc transporter 3 (ZnT3) knockout mice.Behav Brain Res. 2017 Mar 15;321:36-49. doi: 10.1016/j.bbr.2016.12.028. Epub 2016 Dec 21. Behav Brain Res. 2017. PMID: 28012850
-
Cognitive loss in zinc transporter-3 knock-out mice: a phenocopy for the synaptic and memory deficits of Alzheimer's disease?J Neurosci. 2010 Feb 3;30(5):1631-6. doi: 10.1523/JNEUROSCI.5255-09.2010. J Neurosci. 2010. PMID: 20130173 Free PMC article.
-
Trans-synaptic Association of Vesicular Zinc Transporter 3 and Shank3 Supports Synapse-Specific Dendritic Spine Structure and Function in the Mouse Auditory Cortex.J Neurosci. 2024 Jul 10;44(28):e0619242024. doi: 10.1523/JNEUROSCI.0619-24.2024. J Neurosci. 2024. PMID: 38830758 Free PMC article.
-
Zinc transporter 3 (ZnT3) and vesicular zinc in central nervous system function.Neurosci Biobehav Rev. 2017 Sep;80:329-350. doi: 10.1016/j.neubiorev.2017.06.006. Epub 2017 Jun 15. Neurosci Biobehav Rev. 2017. PMID: 28624432 Review.
-
Mechanisms of fear learning and extinction: synaptic plasticity-fear memory connection.Psychopharmacology (Berl). 2019 Jan;236(1):163-182. doi: 10.1007/s00213-018-5104-4. Epub 2018 Nov 10. Psychopharmacology (Berl). 2019. PMID: 30415278 Free PMC article. Review.
Cited by
-
Neuronal signalling of zinc: from detection and modulation to function.Open Biol. 2022 Sep;12(9):220188. doi: 10.1098/rsob.220188. Epub 2022 Sep 7. Open Biol. 2022. PMID: 36067793 Free PMC article. Review.
-
Synergistic activation of dopamine D1 and TrkB receptors mediate gain control of synaptic plasticity in the basolateral amygdala.PLoS One. 2011;6(10):e26065. doi: 10.1371/journal.pone.0026065. Epub 2011 Oct 14. PLoS One. 2011. PMID: 22022509 Free PMC article.
-
Tools and techniques for illuminating the cell biology of zinc.Biochim Biophys Acta Mol Cell Res. 2021 Jan;1868(1):118865. doi: 10.1016/j.bbamcr.2020.118865. Epub 2020 Sep 24. Biochim Biophys Acta Mol Cell Res. 2021. PMID: 32980354 Free PMC article. Review.
-
Cognitive decline due to excess synaptic Zn(2+) signaling in the hippocampus.Front Aging Neurosci. 2014 Feb 27;6:26. doi: 10.3389/fnagi.2014.00026. eCollection 2014. Front Aging Neurosci. 2014. PMID: 24578691 Free PMC article. Review.
-
Cell-type-specific enhancement of deviance detection by synaptic zinc in the mouse auditory cortex.Proc Natl Acad Sci U S A. 2024 Oct;121(40):e2405615121. doi: 10.1073/pnas.2405615121. Epub 2024 Sep 23. Proc Natl Acad Sci U S A. 2024. PMID: 39312661 Free PMC article.
References
-
- Armony JL, Servan-Schreiber D, Romanski LM, Cohen JD, LeDoux JE 1997. Stimulus generalization of fear responses: Effects of auditory cortex lesions in a computational model and in rats. Cereb Cortex 7: 157–165 - PubMed
-
- Bucci DJ, Phillips RG, Burwell RD 2000. Contributions of postrhinal and perirhinal cortex to contextual information processing. Behav Neurosci 114: 882–894 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
