IgG1 is pathogenic in Leishmania mexicana infection
- PMID: 21037092
- PMCID: PMC2989177
- DOI: 10.4049/jimmunol.1002484
IgG1 is pathogenic in Leishmania mexicana infection
Abstract
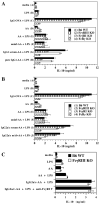
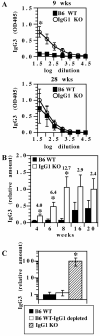
There are >2 million new cases of leishmaniasis annually, and no effective vaccine has been developed to prevent infection. In murine infection, Leishmania mexicana, which lives intracellularly in host macrophages, has developed pathways to hijack host IgG to induce a suppressive IL-10 response through FcγRs, the cell-surface receptors for IgG. To guide vaccine development away from detrimental Ab responses, which can accompany attempts to induce cell-mediated immunity, it is crucial to know which isotypes of IgG are pathogenic in this infection. We found that IgG1 and IgG2a/c induce IL-10 from macrophages in vitro equally well but through different FcγR subtypes: IgG1 through FcγRIII and IgG2a/c through FcγRI primarily, but also through FcγRIII. In sharp contrast, mice lacking IgG1 develop earlier and stronger IgG2a/c, IgG3, and IgM responses to L. mexicana infection and yet are more resistant to the infection. Thus, IgG1, but not IgG2a/c or IgG3, is pathogenic in vivo, in agreement with prior studies indicating that FcγRIII is required for chronic disease. This calls into question the assumption that macrophages, which should secrete IL-10 in response to IgG1 and IgG2a/c immune complexes, are the most important source of IL-10 generated by IgG-FcγR engagement in L. mexicana infection. Further investigations are required to better determine the cell type responsible for this immunosuppressive FcγRIII-induced IL-10 pathway and whether IgG2a/c is protective.
Figures

Similar articles
-
FcgammaRIII mediates immunoglobulin G-induced interleukin-10 and is required for chronic Leishmania mexicana lesions.Infect Immun. 2008 Feb;76(2):623-31. doi: 10.1128/IAI.00316-07. Epub 2007 Dec 10. Infect Immun. 2008. PMID: 18070890 Free PMC article.
-
Interleukin-10 from T cells, but not macrophages and granulocytes, is required for chronic disease in Leishmania mexicana infection.Infect Immun. 2015 Apr;83(4):1366-71. doi: 10.1128/IAI.02909-14. Epub 2015 Jan 20. Infect Immun. 2015. PMID: 25605773 Free PMC article.
-
A detrimental role for IgG and FcgammaR in Leishmania mexicana infection.Immunol Res. 2008;42(1-3):197-209. doi: 10.1007/s12026-008-8074-5. Immunol Res. 2008. PMID: 19002608 Review.
-
Leishmania mexicana infection induces IgG to parasite surface glycoinositol phospholipids that can induce IL-10 in mice and humans.PLoS Negl Trop Dis. 2013 May 9;7(5):e2224. doi: 10.1371/journal.pntd.0002224. Print 2013. PLoS Negl Trop Dis. 2013. PMID: 23675550 Free PMC article.
-
Immune response to Leishmania mexicana: the host-parasite relationship.Pathog Dis. 2020 Nov 11;78(8):ftaa060. doi: 10.1093/femspd/ftaa060. Pathog Dis. 2020. PMID: 33016312 Review.
Cited by
-
Abnormal B-Cell Subset and Blimp-1-Mediated Humoral Responses Associated With Visceral Leishmaniasis Pathogenesis.Am J Trop Med Hyg. 2019 Apr;100(4):816-821. doi: 10.4269/ajtmh.18-0350. Am J Trop Med Hyg. 2019. PMID: 30793688 Free PMC article.
-
A canine model of experimental infection with Leishmania (L.) mexicana.Parasit Vectors. 2014 Aug 9;7:361. doi: 10.1186/1756-3305-7-361. Parasit Vectors. 2014. PMID: 25108307 Free PMC article.
-
Interleukin-4 Responsive Dendritic Cells Are Dispensable to Host Resistance Against Leishmania mexicana Infection.Front Immunol. 2022 Jan 28;12:759021. doi: 10.3389/fimmu.2021.759021. eCollection 2021. Front Immunol. 2022. PMID: 35154068 Free PMC article.
-
Comment on "Regulation of immunity during visceral Leishmania infection" and further discussions about the role of antibodies in infections with Leishmania.Parasit Vectors. 2016 Jul 7;9(1):386. doi: 10.1186/s13071-016-1669-0. Parasit Vectors. 2016. PMID: 27387545 Free PMC article.
-
Protective Efficacy of the Epitope-Conjugated Antigen N-Tc52/TSkb20 in Mitigating Trypanosoma cruzi Infection through CD8+ T-Cells and IFNγ Responses.Vaccines (Basel). 2024 Jun 4;12(6):621. doi: 10.3390/vaccines12060621. Vaccines (Basel). 2024. PMID: 38932350 Free PMC article.
References
-
- World Health Organization Leishmaniasis. [Accessed: October 14, 2010]. 2005. Avalable at: http://www.who.int/leishmaniasis/en/
-
- World Health Organization World Health Report: Reducing Risks, Promoting Healthy Life. [Accessed: October 14, 2010]. 2002. Available at: http://www.who.int/whr/2002/en/whr02_en.pdf.
-
- Bryceson AD, Chulay JD, Ho M, Mugambii M, Were JB, Muigai R, Chunge C, Gachihi G, Meme J, Anabwani G. Visceral leishmaniasis unresponsive to antimonial drugs. I. Clinical and immunological studies. Trans. R. Soc. Trop. Med. Hyg. 1985;79:700–704. e. al. - PubMed
-
- Sundar S, More DK, Singh MK, Singh VP, Sharma S, Makharia A, Kumar PC, Murray HW. Failure of pentavalent antimony in visceral leishmaniasis in India: report from the center of the Indian epidemic. Clin. Infect. Dis. 2000;31:1104–1107. - PubMed
-
- Soto J, Toledo J, Vega J, Berman J. Short report: efficacy of pentavalent antimony for treatment of colombian cutaneous leishmaniasis. Am. J. Trop. Med. Hyg. 2005;72:421–422. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

