Analysis of distinct short and prolonged components in rebound spiking of deep cerebellar nucleus neurons
- PMID: 21039958
- PMCID: PMC3058674
- DOI: 10.1111/j.1460-9568.2010.07408.x
Analysis of distinct short and prolonged components in rebound spiking of deep cerebellar nucleus neurons
Abstract
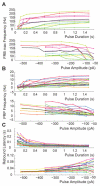
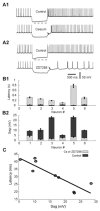
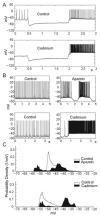
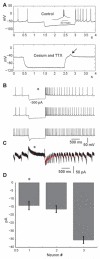
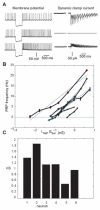
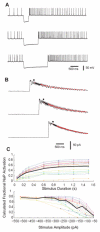
Deep cerebellar nucleus (DCN) neurons show pronounced post-hyperpolarization rebound burst behavior, which may contribute significantly to responses to strong inhibitory inputs from cerebellar cortical Purkinje cells. Thus, rebound behavior could importantly shape the output from the cerebellum. We used whole-cell recordings in brain slices to characterize DCN rebound properties and their dependence on hyperpolarization duration and depth. We found that DCN rebounds showed distinct fast and prolonged components, with different stimulus dependence and different underlying currents. The initial depolarization leading into rebound spiking was carried by hyperpolarization-activated cyclic nucleotide-gated current, and variable expression of this current could lead to a control of rebound latency. The ensuing fast rebound burst was due to T-type calcium current, as previously described. It was highly variable between cells in strength, and could be expressed fully after short periods of hyperpolarization. In contrast, a subsequent prolonged rebound component required longer and deeper periods of hyperpolarization before it was fully established. We found using voltage-clamp and dynamic-clamp analyses that a slowly inactivating persistent sodium current fits the conductance underlying this prolonged rebound component, resulting in spike rate increases over several seconds. Overall, our results demonstrate that multiphasic DCN rebound properties could be elicited differentially by different levels of Purkinje cell activation, and thus create a rich repertoire of potential rebound dynamics in the cerebellar control of motor timing.
© 2010 The Authors. European Journal of Neuroscience © 2010 Federation of European Neuroscience Societies and Blackwell Publishing Ltd.
Figures


References
-
- Aizenman CD, Huang EJ, Linden DJ. Morphological correlates of intrinsic electrical excitability in neurons of the deep cerebellar nuclei. J Neurophysiol. 2003;89:1738–1747. - PubMed
-
- Aizenman CD, Linden DJ. Regulation of the rebound depolarization and spontaneous firing patterns of deep nuclear neurons in slices of rat cerebellum. J Neurophysiol. 1999;82:1697–1709. - PubMed
-
- Aizenman CD, Manis PB, Linden DJ. Polarity of long-term synaptic gain change is related to postsynaptic spike firing at a cerebellar inhibitory synapse. Neuron. 1998;21:827–835. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

