Agonist-induced localization of Gq-coupled receptors and G protein-gated inwardly rectifying K+ (GIRK) channels to caveolae determines receptor specificity of phosphatidylinositol 4,5-bisphosphate signaling
- PMID: 21041306
- PMCID: PMC3009900
- DOI: 10.1074/jbc.M110.153312
Agonist-induced localization of Gq-coupled receptors and G protein-gated inwardly rectifying K+ (GIRK) channels to caveolae determines receptor specificity of phosphatidylinositol 4,5-bisphosphate signaling
Abstract
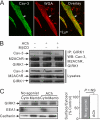
G protein-gated inwardly rectifying K(+) (GIRK) channels are parasympathetic effectors in cardiac myocytes that act as points of integration of signals from diverse pathways. Neurotransmitters and hormones acting on the Gq protein regulate GIRK channels by phosphatidylinositol 4,5-bisphosphate (PIP(2)) depletion. In previous studies, we found that endothelin-1, but not bradykinin, inhibited GIRK channels, even though both of them hydrolyze PIP(2) in cardiac myocytes, showing receptor specificity. The present study assessed whether the spatial organization of the PIP(2) signal into caveolar microdomains underlies the specificity of PIP(2)-mediated signaling. Using biochemical analysis, we examined the localization of GIRK and Gq protein-coupled receptors (GqPCRs) in mouse atrial myocytes. Agonist stimulation induced a transient co-localization of GIRK channels with endothelin receptors in the caveolae, excluding bradykinin receptors. Such redistribution was eliminated by caveolar disruption with methyl-β-cyclodextrin (MβCD). Patch clamp studies showed that the specific response of GIRK channels to GqPCR agonists was abolished by MβCD, indicating the functional significance of the caveolae-dependent spatial organization. To assess whether low PIP(2) mobility is essential for PIP(2)-mediated signaling, we blocked the cytoskeletal restriction of PIP(2) diffusion by latrunculin B. This abolished the GIRK channel regulation by GqPCRs without affecting their targeting to caveolae. These data suggest that without the hindered diffusion of PIP(2) from microdomains, PIP(2) loses its signaling efficacy. Taken together, these data suggest that specific targeting combined with restricted diffusion of PIP(2) allows the PIP(2) signal to be compartmentalized to the targets localized closely to the GqPCRs, enabling cells to discriminate between identical PIP(2) signaling that is triggered by different receptors.
Figures

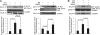



References
-
- Sharifov O. F., Fedorov V. V., Beloshapko G. G., Glukhov A. V., Yushmanova A. V., Rosenshtraukh L. V. (2004) J. Am. Coll. Cardiol. 43, 483–490 - PubMed
-
- Kolettis T. M., Kyriakides Z. S., Zygalaki E., Kyrzopoulos S., Kaklamanis L., Nikolaou N., Lianidou E. S., Kremastinos D. T. (2008) J. Interv. Card. Electrophysiol. 21, 203–208 - PubMed
-
- Tsai C. T., Lai L. P., Lin J. L., Chiang F. T., Hwang J. J., Ritchie M. D., Moore J. H., Hsu K. L., Tseng C. D., Liau C. S., Tseng Y. Z. (2004) Circulation 109, 1640–1646 - PubMed
-
- Kitakaze M., Node K., Takashima S., Minamino T., Kuzuya T., Hori M. (2000) Hypertens. Res. 23, 253–259 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

