Coevolution of TH1, TH2, and TH17 responses during repeated pulmonary exposure to Aspergillus fumigatus conidia
- PMID: 21041495
- PMCID: PMC3019910
- DOI: 10.1128/IAI.00508-10
Coevolution of TH1, TH2, and TH17 responses during repeated pulmonary exposure to Aspergillus fumigatus conidia
Abstract
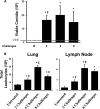
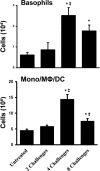
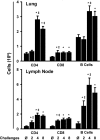
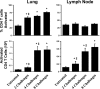
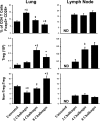
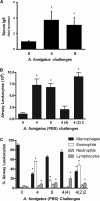
Aspergillus fumigatus, a ubiquitous airborne fungus, can cause invasive infection in immunocompromised individuals but also triggers allergic bronchopulmonary aspergillosis in a subset of otherwise healthy individuals repeatedly exposed to the organism. This study addresses a critical gap in our understanding of the immunoregulation in response to repeated exposure to A. fumigatus conidia. C57BL/6 mice were challenged intranasally with A. fumigatus conidia weekly, and leukocyte composition, activation, and cytokine production were examined after two, four, and eight challenges. Approximately 99% of A. fumigatus conidia were cleared within 24 h after inoculation, and repeated exposure to A. fumigatus conidia did not result in hyphal growth or accumulation of conidia with time. After 2 challenges, there was an early influx of neutrophils and regulatory T (T(reg)) cells into the lungs but minimal inflammation. Repeated exposure promoted sustained expansion of the draining lymph nodes, while the influx of eosinophils and other myeloid cells into the lungs peaked after four exposures and then decreased despite continued A. fumigatus challenges. Goblet cell metaplasia and low-level fibrosis were evident during the response. Repeated exposure to A. fumigatus conidia induced T cell activation in the lungs and the codevelopment by four exposures of T(H)1, T(H)2, and T(H)17 responses in the lungs, which were maintained through eight exposures. Changes in CD4 T cell polarization or T(reg) numbers did not account for the reduction in myeloid cell numbers later in the response, suggesting a non-T-cell regulatory pathway involved in dampening inflammation during repeated exposure to A. fumigatus conidia.
Figures


References
-
- Aebischer, C. C., U. Frey, and M. H. Schoni. 2002. Hypersensitivity pneumonitis in a five-year-old boy: an unusual antigen source. Pediatr. Pulmonol. 33:77-78. - PubMed
-
- Armstrong-James, D. P., S. A. Turnbull, I. Teo, J. Stark, N. J. Rogers, T. R. Rogers, E. Bignell, and K. Haynes. 2009. Impaired interferon-gamma responses, increased interleukin-17 expression, and a tumor necrosis factor-alpha transcriptional program in invasive aspergillosis. J. Infect. Dis. 200:1341-1351. - PubMed
-
- Bartlett, N. W., R. P. Walton, M. R. Edwards, J. Aniscenko, G. Caramori, J. Zhu, N. Glanville, K. J. Choy, P. Jourdan, J. Burnet, T. J. Tuthill, M. S. Pedrick, M. J. Hurle, C. Plumpton, N. A. Sharp, J. N. Bussell, D. M. Swallow, J. Schwarze, B. Guy, J. W. Almond, P. K. Jeffery, C. M. Lloyd, A. Papi, R. A. Killington, D. J. Rowlands, E. D. Blair, N. J. Clarke, and S. L. Johnston. 2008. Mouse models of rhinovirus-induced disease and exacerbation of allergic airway inflammation. Nat. Med. 14:199-204. - PMC - PubMed
-
- Bellocchio, S., S. Moretti, K. Perruccio, F. Fallarino, S. Bozza, C. Montagnoli, P. Mosci, G. B. Lipford, L. Pitzurra, and L. Romani. 2004. TLRs govern neutrophil activity in aspergillosis. J. Immunol. 173:7406-7415. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

