Hemoglobin redux: combining neutron and X-ray diffraction with mass spectrometry to analyse the quaternary state of oxidized hemoglobins
- PMID: 21041946
- PMCID: PMC2967423
- DOI: 10.1107/S090744491002545X
Hemoglobin redux: combining neutron and X-ray diffraction with mass spectrometry to analyse the quaternary state of oxidized hemoglobins
Abstract
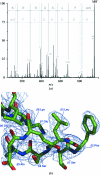
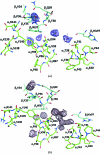
Improvements in neutron diffraction instrumentation are affording the opportunity to re-examine the structures of vertebrate hemoglobins and to interrogate proton and solvent position changes between the different quaternary states of the protein. For hemoglobins of unknown primary sequence, structural studies of cyanomethemoglobin (CNmetHb) are being used to help to resolve sequence ambiguity in the mass spectra. These studies have also provided additional structural evidence for the involvement of oxidized hemoglobin in the process of erythrocyte senescence. X-ray crystal studies of Tibetan snow leopard CNmetHb have shown that this protein crystallizes in the B state, a structure with a more open dyad, which possibly has relevance to RBC band 3 protein binding and erythrocyte senescence. R-state equine CNmetHb crystal studies elaborate the solvent differences in the switch and hinge region compared with a human deoxyhemoglobin T-state neutron structure. Lastly, comparison of histidine protonation between the T and R state should enumerate the Bohr-effect protons.
Figures


Similar articles
-
Visualizing the Bohr effect in hemoglobin: neutron structure of equine cyanomethemoglobin in the R state and comparison with human deoxyhemoglobin in the T state.Acta Crystallogr D Struct Biol. 2016 Jul;72(Pt 7):892-903. doi: 10.1107/S2059798316009049. Epub 2016 Jun 28. Acta Crystallogr D Struct Biol. 2016. PMID: 27377386 Free PMC article.
-
Preliminary neutron and X-ray crystallographic studies of equine cyanomethemoglobin.Acta Crystallogr Sect F Struct Biol Cryst Commun. 2010 Apr 1;66(Pt 4):474-7. doi: 10.1107/S1744309110007840. Epub 2010 Mar 31. Acta Crystallogr Sect F Struct Biol Cryst Commun. 2010. PMID: 20383026 Free PMC article.
-
A new relaxed state in horse methemoglobin characterized by crystallographic studies.Proteins. 2005 Aug 15;60(3):547-51. doi: 10.1002/prot.20510. Proteins. 2005. PMID: 15887226
-
Metalloprotein catalysis: structural and mechanistic insights into oxidoreductases from neutron protein crystallography.Acta Crystallogr D Struct Biol. 2021 Oct 1;77(Pt 10):1251-1269. doi: 10.1107/S2059798321009025. Epub 2021 Sep 27. Acta Crystallogr D Struct Biol. 2021. PMID: 34605429 Free PMC article. Review.
-
The use of neutron scattering to determine the functional structure of glycoside hydrolase.Curr Opin Struct Biol. 2016 Oct;40:54-61. doi: 10.1016/j.sbi.2016.07.014. Epub 2016 Aug 3. Curr Opin Struct Biol. 2016. PMID: 27494120 Review.
References
-
- Altland, P. D. & Brace, K. C. (1962). Am. J. Physiol.201, 1188–1190.
-
- Berenbrink, M. (2006). Resp. Physiol. Neurobiol.154, 165–184. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

