The FLARE intraoperative near-infrared fluorescence imaging system: a first-in-human clinical trial in perforator flap breast reconstruction
- PMID: 21042103
- PMCID: PMC2974179
- DOI: 10.1097/PRS.0b013e3181f059c7
The FLARE intraoperative near-infrared fluorescence imaging system: a first-in-human clinical trial in perforator flap breast reconstruction
Abstract
Background: The ability to determine flap perfusion in reconstructive surgery is still primarily based on clinical examination. In this study, the authors demonstrate the use of an intraoperative, near-infrared fluorescence imaging system for evaluation of perforator location and flap perfusion.
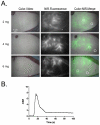
Methods: Indocyanine green was injected intravenously in six breast cancer patients undergoing a deep inferior epigastric perforator flap breast reconstruction after mastectomy. Three dose levels of indocyanine green were assessed using the fluorescence-assisted resection and exploration (FLARE) imaging system. This system uses light-emitting diodes for fluorescence excitation, which is different from current commercially available systems. In this pilot study, the operating surgeons were blinded to the imaging results.
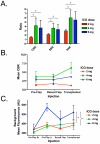
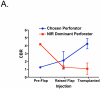
Results: Use of the FLARE system was successful in all six study subjects, with no complications or sequelae. Among the three dose levels, 4 mg per injection resulted in the highest observed contrast-to-background ratio, signal-to-background ratio, and signal-to-noise ratio. However, because of small sample size, the authors did not have sufficient power to detect statistical significance for these pairwise comparisons at the multiple-comparison adjusted type I error of 0.017. Six milligrams per injection provided a similar contrast-to-background ratio but also a higher residual background signal.
Conclusion: Based on this pilot study, the authors conclude that near-infrared assessment of perforator flap breast reconstruction is feasible with a light-emitting diode-based system, and that a dose of 4 mg of indocyanine green per injection yields the best observed contrast-to-background ratio compared with a dose of 2 or 6 mg for assessment of flap perfusion.
Trial registration: ClinicalTrials.gov NCT00952107.
Figures


Similar articles
-
Intraoperative perfusion mapping with laser-assisted indocyanine green imaging can predict and prevent complications in immediate breast reconstruction.Plast Reconstr Surg. 2010 Apr;125(4):1065-1073. doi: 10.1097/PRS.0b013e3181d17f80. Plast Reconstr Surg. 2010. PMID: 20335859
-
Intraoperative near-infrared fluorescence imaging in perforator flap reconstruction: current research and early clinical experience.J Reconstr Microsurg. 2010 Jan;26(1):59-65. doi: 10.1055/s-0029-1244805. Epub 2009 Dec 21. J Reconstr Microsurg. 2010. PMID: 20027541 Free PMC article. Clinical Trial.
-
The ability of intra-operative perfusion mapping with laser-assisted indocyanine green angiography to predict mastectomy flap necrosis in breast reconstruction: a prospective trial.J Plast Reconstr Aesthet Surg. 2014 Apr;67(4):449-55. doi: 10.1016/j.bjps.2013.12.040. Epub 2013 Dec 31. J Plast Reconstr Aesthet Surg. 2014. PMID: 24507962
-
Use of laser-assisted indocyanine green angiography in breast reconstruction: Systematic review and meta-analysis.J Surg Oncol. 2020 Apr;121(5):759-765. doi: 10.1002/jso.25782. Epub 2019 Nov 26. J Surg Oncol. 2020. PMID: 31773735
-
Clinical and Cost-Effectiveness of Intraoperative Flap Perfusion Assessment With Indocyanine Green Fluorescence Angiography in Breast and Head and Neck Reconstructions: A Systematic Review and Meta-Analysis.Microsurgery. 2024 Nov;44(8):e31250. doi: 10.1002/micr.31250. Microsurgery. 2024. PMID: 39449167
Cited by
-
Direct Administration of Nerve-Specific Contrast to Improve Nerve Sparing Radical Prostatectomy.Theranostics. 2017 Jan 7;7(3):573-593. doi: 10.7150/thno.17433. eCollection 2017. Theranostics. 2017. PMID: 28255352 Free PMC article.
-
Molecular imaging of breast cancer: present and future directions.Front Chem. 2014 Dec 18;2:112. doi: 10.3389/fchem.2014.00112. eCollection 2014. Front Chem. 2014. PMID: 25566530 Free PMC article. Review.
-
Indocyanine Green-Loaded PLGA Nanoparticles Conjugated with Hyaluronic Acid Improve Target Specificity in Cervical Cancer Tumors.Yonsei Med J. 2021 Nov;62(11):1042-1051. doi: 10.3349/ymj.2021.62.11.1042. Yonsei Med J. 2021. PMID: 34672138 Free PMC article.
-
Real-time fluorescence image-guided oncologic surgery.Adv Cancer Res. 2014;124:171-211. doi: 10.1016/B978-0-12-411638-2.00005-7. Adv Cancer Res. 2014. PMID: 25287689 Free PMC article. Review.
-
Prediction of Skin Necrosis after Mastectomy for Breast Cancer Using Indocyanine Green Angiography Imaging.Plast Reconstr Surg Glob Open. 2017 Apr 21;5(4):e1321. doi: 10.1097/GOX.0000000000001321. eCollection 2017 Apr. Plast Reconstr Surg Glob Open. 2017. PMID: 28507876 Free PMC article.
References
-
- Giunta RE, Geisweid A, Feller AM. The value of preoperative Doppler sonography for planning free perforator flaps. Plast Reconstr Surg. 2000;105:2381–2386. - PubMed
-
- Masia J, Clavero JA, Larranaga JR, et al. Multidetector-row computed tomography in the planning of abdominal perforator flaps. J Plast Reconstr Aesthet Surg. 2006;59:594–599. - PubMed
-
- Rozen WM, Stella DL, Phillips TJ, et al. Magnetic resonance angiography in the preoperative planning of DIEA perforator flaps. Plast Reconstr Surg. 2008;122:222e–223e. - PubMed
-
- Saint-Cyr M, Schaverien M, Arbique G, et al. Three- and four-dimensional computed tomographic angiography and venography for the investigation of the vascular anatomy and perfusion of perforator flaps. Plast Reconstr Surg. 2008;121:772–780. - PubMed
-
- Mathes DW, Neligan PC. Current techniques in preoperative imaging for abdomen-based perforator flap microsurgical breast reconstruction. J Reconstr Microsurg. 2010;26:3–10. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

