Oligomers of a 5-carboxy-methanopyrrolidine β-amino acid. A search for order
- PMID: 21043445
- PMCID: PMC2993784
- DOI: 10.1021/ol1022917
Oligomers of a 5-carboxy-methanopyrrolidine β-amino acid. A search for order
Abstract
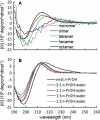
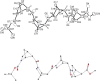
CD spectra for homooligomers (n = 4, 6, 8) of (1S,4R,5R)-5-syn-carboxy-2-azabicyclo[2.1.1]hexane (MPCA), a methano-bridged pyrrolidine β-carboxylic acid, suggest an ordered secondary structure. Even in the absence of internal hydrogen bonding, solution NMR, X-ray, and in silico analyses of the tetramer are indicative of conformations with trans-amides and C(5)-amide-carbonyls oriented toward the C(4) bridgehead. This highly constrained β-amino acid could prove useful in the ongoing development of well-defined foldamers.
Figures
References
-
- Kakinoki S, Hirano Y, Oka M. Polymer Bulletin. 2005;53:109–115.
- Cowan PM, McGavin S. Nature. 1955;176:501–503. - PubMed
- Traub W, Shmueli U. Nature. 1963;198:1165–1166.
-
- Abele S, Vogtli K, Seebach D. Helv. Chim. Acta. 1999;82:1539–1558.
-
It was known that peptoids form ordered secondary structures; see ref. 18–21 therein.
-
- Huck BR, Langenhan JM, Gellman SH. Org. Lett. 1999;1:1717–1720. - PubMed
-
- Sandvoss LM, Carlson HA. J. Am. Chem. Soc. 2003;125:15855–15862. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

