Evidence that αC region is origin of low modulus, high extensibility, and strain stiffening in fibrin fibers
- PMID: 21044602
- PMCID: PMC2965937
- DOI: 10.1016/j.bpj.2010.08.060
Evidence that αC region is origin of low modulus, high extensibility, and strain stiffening in fibrin fibers
Abstract
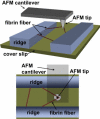
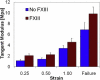
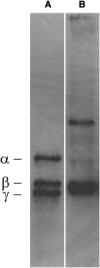
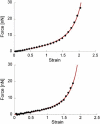
Fibrin fibers form the structural scaffold of blood clots and perform the mechanical task of stemming blood flow. Several decades of investigation of fibrin fiber networks using macroscopic techniques have revealed remarkable mechanical properties. More recently, the microscopic origins of fibrin's mechanics have been probed through direct measurements on single fibrin fibers and individual fibrinogen molecules. Using a nanomanipulation system, we investigated the mechanical properties of individual fibrin fibers. The fibers were stretched with the atomic force microscope, and stress-versus-strain data was collected for fibers formed with and without ligation by the activated transglutaminase factor XIII (FXIIIa). We observed that ligation with FXIIIa nearly doubled the stiffness of the fibers. The stress-versus-strain behavior indicates that fibrin fibers exhibit properties similar to other elastomeric biopolymers. We propose a mechanical model that fits our observed force extension data, is consistent with the results of the ligation data, and suggests that the large observed extensibility in fibrin fibers is mediated by the natively unfolded regions of the molecule. Although some models attribute fibrin's force-versus-extension behavior to unfolding of structured regions within the monomer, our analysis argues that these models are inconsistent with the measured extensibility and elastic modulus.
Copyright © 2010 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures




References
-
- Weisel J.W. The mechanical properties of fibrin for basic scientists and clinicians. Biophys. Chem. 2004;112:267–276. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

