Partial agonists of the α3β4* neuronal nicotinic acetylcholine receptor reduce ethanol consumption and seeking in rats
- PMID: 21048701
- PMCID: PMC3055681
- DOI: 10.1038/npp.2010.191
Partial agonists of the α3β4* neuronal nicotinic acetylcholine receptor reduce ethanol consumption and seeking in rats
Abstract
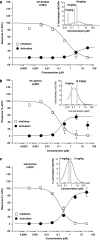
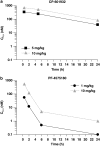
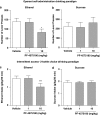
Alcohol use disorders (AUDs) impact millions of individuals and there remain few effective treatment strategies. Despite evidence that neuronal nicotinic acetylcholine receptors (nAChRs) have a role in AUDs, it has not been established which subtypes of the nAChR are involved. Recent human genetic association studies have implicated the gene cluster CHRNA3-CHRNA5-CHRNB4 encoding the α3, α5, and β4 subunits of the nAChR in susceptibility to develop nicotine and alcohol dependence; however, their role in ethanol-mediated behaviors is unknown due to the lack of suitable and selective research tools. To determine the role of the α3, and β4 subunits of the nAChR in ethanol self-administration, we developed and characterized high-affinity partial agonists at α3β4 nAChRs, CP-601932, and PF-4575180. Both CP-601932 and PF-4575180 selectively decrease ethanol but not sucrose consumption and operant self-administration following long-term exposure. We show that the functional potencies of CP-601932 and PF-4575180 at α3β4 nAChRs correlate with their unbound rat brain concentrations, suggesting that the effects on ethanol self-administration are mediated via interaction with α3β4 nAChRs. Also varenicline, an approved smoking cessation aid previously shown to decrease ethanol consumption and seeking in rats and mice, reduces ethanol intake at unbound brain concentrations that allow functional interactions with α3β4 nAChRs. Furthermore, the selective α4β2(*) nAChR antagonist, DHβE, did not reduce ethanol intake. Together, these data provide further support for the human genetic association studies, implicating CHRNA3 and CHRNB4 genes in ethanol-mediated behaviors. CP-601932 has been shown to be safe in humans and may represent a potential novel treatment for AUDs.
Figures


Comment in
-
Neuronal nicotinic acetylcholine receptors: emerging therapeutic targets for alcoholism. Commentary.CNS Neurol Disord Drug Targets. 2011 Aug;10(5):526. doi: 10.2174/187152711796234961. CNS Neurol Disord Drug Targets. 2011. PMID: 21631405 No abstract available.
References
-
- Balcombe JP, Barnard ND, Sandusky C. Laboratory routines cause animal stress. Contemp Top Lab Anim Sci. 2004;43:42–51. - PubMed
-
- Blomqvist O, Soderpalm B, Engel JA. Ethanol-induced locomotor activity: involvement of central nicotinic acetylcholine receptors. Brain Res Bull. 1992;29:173–178. - PubMed
-
- Cahill K, Stead L, Lancaster T. A preliminary benefit-risk assessment of varenicline in smoking cessation. Drug Saf. 2009;32:119–135. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

