Integrating carbon-halogen bond formation into medicinal plant metabolism
- PMID: 21048708
- PMCID: PMC3058899
- DOI: 10.1038/nature09524
Integrating carbon-halogen bond formation into medicinal plant metabolism
Abstract
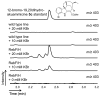
Halogenation, which was once considered a rare occurrence in nature, has now been observed in many natural product biosynthetic pathways. However, only a small fraction of halogenated compounds have been isolated from terrestrial plants. Given the impact that halogenation can have on the biological activity of natural products, we reasoned that the introduction of halides into medicinal plant metabolism would provide the opportunity to rationally bioengineer a broad variety of novel plant products with altered, and perhaps improved, pharmacological properties. Here we report that chlorination biosynthetic machinery from soil bacteria can be successfully introduced into the medicinal plant Catharanthus roseus (Madagascar periwinkle). These prokaryotic halogenases function within the context of the plant cell to generate chlorinated tryptophan, which is then shuttled into monoterpene indole alkaloid metabolism to yield chlorinated alkaloids. A new functional group-a halide-is thereby introduced into the complex metabolism of C. roseus, and is incorporated in a predictable and regioselective manner onto the plant alkaloid products. Medicinal plants, despite their genetic and developmental complexity, therefore seem to be a viable platform for synthetic biology efforts.
Figures


Comment in
-
Chemical biology: Synthetic metabolism goes green.Nature. 2010 Nov 18;468(7322):380-1. doi: 10.1038/468380a. Nature. 2010. PMID: 21085165 No abstract available.
Similar articles
-
Chemical biology: Synthetic metabolism goes green.Nature. 2010 Nov 18;468(7322):380-1. doi: 10.1038/468380a. Nature. 2010. PMID: 21085165 No abstract available.
-
Transformation with tryptophan halogenase genes leads to the production of new chlorinated alkaloid metabolites by a medicinal plant.Chembiochem. 2011 Mar 21;12(5):681-3. doi: 10.1002/cbic.201100016. Epub 2011 Feb 23. Chembiochem. 2011. PMID: 21344584 No abstract available.
-
Reengineering a tryptophan halogenase to preferentially chlorinate a direct alkaloid precursor.J Am Chem Soc. 2011 Dec 7;133(48):19346-9. doi: 10.1021/ja2089348. Epub 2011 Nov 11. J Am Chem Soc. 2011. PMID: 22050348
-
Biosynthetic pathway of terpenoid indole alkaloids in Catharanthus roseus.Front Med. 2014 Sep;8(3):285-93. doi: 10.1007/s11684-014-0350-2. Epub 2014 Aug 26. Front Med. 2014. PMID: 25159992 Review.
-
Terpenoid indole alkaloid biosynthesis in Catharanthus roseus: effects and prospects of environmental factors in metabolic engineering.Biotechnol Lett. 2021 Nov;43(11):2085-2103. doi: 10.1007/s10529-021-03179-x. Epub 2021 Sep 25. Biotechnol Lett. 2021. PMID: 34564757 Free PMC article. Review.
Cited by
-
A halogen budget of the bulk silicate Earth points to a history of early halogen degassing followed by net regassing.Proc Natl Acad Sci U S A. 2021 Dec 21;118(51):e2116083118. doi: 10.1073/pnas.2116083118. Proc Natl Acad Sci U S A. 2021. PMID: 34916297 Free PMC article.
-
A Leader-Guided Substrate Tolerant RiPP Brominase Allows Suzuki-Miyaura Cross-Coupling Reactions for Peptides and Proteins.Biochemistry. 2023 Jun 20;62(12):1838-1843. doi: 10.1021/acs.biochem.3c00222. Epub 2023 Jun 5. Biochemistry. 2023. PMID: 37272553 Free PMC article.
-
Deploying Microbial Synthesis for Halogenating and Diversifying Medicinal Alkaloid Scaffolds.Front Bioeng Biotechnol. 2020 Oct 23;8:594126. doi: 10.3389/fbioe.2020.594126. eCollection 2020. Front Bioeng Biotechnol. 2020. PMID: 33195162 Free PMC article. Review.
-
Functional characterization of a catalytically promiscuous tryptophan decarboxylase from camptothecin-producing Camptotheca acuminata.Front Plant Sci. 2022 Aug 18;13:987348. doi: 10.3389/fpls.2022.987348. eCollection 2022. Front Plant Sci. 2022. PMID: 36061783 Free PMC article.
-
Identifying enriched drug fragments as possible candidates for metabolic engineering.BMC Med Genomics. 2016 Aug 10;9 Suppl 2(Suppl 2):46. doi: 10.1186/s12920-016-0205-6. BMC Med Genomics. 2016. PMID: 27510651 Free PMC article.
References
-
- Neumann CS, Fujimori DG, Walsh CT. Halogenation strategies in natural products biosynthesis. Chem Biol. 2008;15:99–109. - PubMed
-
- Gribble GW. The diversity of naturally produced organohalogens. Chemosphere. 2003;52:289–297. - PubMed
-
- Vaillancourt FH, Yeh E, Vosburg DA, Garneau-Tsodiova S, Walsh CT. Nature’s inventory of halogenation catalysts: oxidative strategies predominate. Chem Rev. 2006;106:3364–3378. - PubMed
-
- van Peé KH, Patallo EP. Flavin-dependent halogenases involved in secondary metabolism in bacteria. Appl Microbiol Biotechnol. 2006;70:631–641. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

