Inactivation of lipid glyceryl ester metabolism in human THP1 monocytes/macrophages by activated organophosphorus insecticides: role of carboxylesterases 1 and 2
- PMID: 21049984
- PMCID: PMC4065498
- DOI: 10.1021/tx1002194
Inactivation of lipid glyceryl ester metabolism in human THP1 monocytes/macrophages by activated organophosphorus insecticides: role of carboxylesterases 1 and 2
Abstract
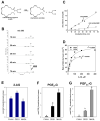
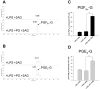
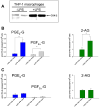
Carboxylesterases (CES) have important roles in pesticide and drug metabolism and contribute to the clearance of ester-containing xenobiotics in mammals. Tissues with the highest levels of CES expression are the liver and small intestine. In addition to xenobiotics, CES also harness their broad substrate specificity to hydrolyze endobiotics, such as cholesteryl esters and triacylglycerols. Here, we determined if two human CES isoforms, CES1 and CES2, hydrolyze the endocannabinoids 2-arachidonoylglycerol (2AG) and anandamide (AEA), and two prostaglandin glyceryl esters (PG-Gs), which are formed by COX-mediated oxygenation of 2AG. We show that recombinant CES1 and CES2 efficiently hydrolyze 2AG to arachidonic acid (AA) but not amide-containing AEA. Steady-state kinetic parameters for CES1- and CES2-mediated 2AG hydrolysis were, respectively, kcat, 59 and 43 min(-1); Km, 49 and 46 μM; and kcat/Km, 1.2 and 0.93 μM(-1) min(-1). kcat/Km values are comparable to published values for rat monoacylglycerol lipase (MAGL)-catalyzed 2AG hydrolysis. Furthermore, we show that CES1 and CES2 also efficiently hydrolyze PGE2-G and PGF2α-G. In addition, when cultured human THP1 macrophages were treated with exogenous 2AG or PG-G (10 μM, 1 h), significant quantities of AA or PGs were detected in the culture medium; however, the ability of macrophages to metabolize these compounds was inhibited (60-80%) following treatment with paraoxon, the toxic metabolite of the insecticide parathion. Incubation of THP1 cell lysates with small-molecule inhibitors targeting CES1 (thieno[3,2-e][1]benzothiophene-4,5-dione or JZL184) significantly reduced lipid glyceryl ester hydrolase activities (40-50% for 2AG and 80-95% for PG-Gs). Immunodepletion of CES1 also markedly reduced 2AG and PG-G hydrolase activities. These results suggested that CES1 is in part responsible for the hydrolysis of 2AG and PG-Gs in THP1 cells, although it did not rule out a role for other hydrolases, especially with regard to 2AG metabolism since a substantial portion of its hydrolysis was not inactivated by the inhibitors. An enzyme (Mr 31-32 kDa) of unknown function was detected by serine hydrolase activity profiling of THP1 cells and may be a candidate. Finally, the amounts of in situ generated 2AG and PG-Gs in macrophages were enhanced by treating the cells with bioactive metabolites of OP insecticides. Collectively, the results suggest that in addition to MAGL and fatty-acid amide hydrolase (FAAH), which have both been documented to terminate endocannabinoid signaling, CES may also have a role. Furthermore, since PG-Gs have been shown to possess biological activities in their own right, CES may represent an important enzyme class that regulates their in vivo levels.
Figures






References
-
- Di Marzo V. Targeting the endocannabinoid system: to enhance or reduce? Nat. Rev. Drug Discov. 2008;7:438–455. - PubMed
-
- Piomelli D. The molecular logic of endocannabinoid signalling. Nat. Rev. Neurosci. 2003;4:873–884. - PubMed
-
- Kozak KR, Rowlinson SW, Marnett LJ. Oxygenation of the endocannabinoid, 2-arachidonylglycerol, to glyceryl prostaglandins by cyclooxygenase-2. J. Biol. Chem. 2000;275:33744–33749. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

