DRUG FOCUS: S 18986: A positive allosteric modulator of AMPA-type glutamate receptors pharmacological profile of a novel cognitive enhancer
- PMID: 21050420
- PMCID: PMC6493918
- DOI: 10.1111/j.1755-5949.2009.00088.x
DRUG FOCUS: S 18986: A positive allosteric modulator of AMPA-type glutamate receptors pharmacological profile of a novel cognitive enhancer
Abstract
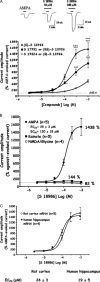
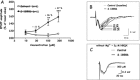
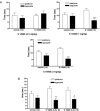
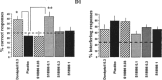
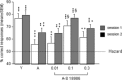
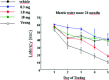
Alpha-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid (AMPA) type glutamate receptors are critical for synaptic plasticity and induction of long-term potentiation (LTP), considered as one of the synaptic mechanisms underlying learning and memory. Positive allosteric modulators of AMPA receptors could provide a therapeutic approach to the treatment of cognitive disorders resulting from aging and/or neurodegenerative diseases, such as Alzheimer disease (AD). Several AMPA potentiators have been described in the last decade, but for the moment their clinical efficacy has not been demonstrated due to the complexity of the target, AMPA receptors, and the difficulty in studying cognition in animals and humans. A better understanding of the mechanism of action of this type of drug remains an important issue, if knowledge of these compounds is to be increased and if this novel therapeutic approach is to be an interesting research area. Among the AMPA potentiators, S 18986 is emerging as a new selective positive allosteric modulator of AMPA-type glutamate receptors. S 18986, as with other positive AMPA receptor modulators, increased induction and maintenance of LTP in the hippocampus as well as the expression of brain-derived neurotrophic factor (BDNF) both in vitro and in vivo. Its cognitive-enhancing properties have been demonstrated in various behavioral models (procedural, spatial, "episodic," working, and relational/declarative memory) in young-adult and aged rodents. It is interesting to note that memory-enhancing effects appeared more robust in middle-aged animals compared with aged ones and in "episodic" and spatial memory tasks. From these results, S 18986 is expected to treat memory deficits associated with early cerebral aging and neurological diseases in elderly people.
© 2010 Blackwell Publishing Ltd.
Conflict of interest statement
None.
Figures





Similar articles
-
AMPA receptor potentiators as cognitive enhancers.IDrugs. 2007 Mar;10(3):185-92. IDrugs. 2007. PMID: 17351873 Review.
-
The AMPA modulator S 18986 improves declarative and working memory performances in aged mice.Behav Pharmacol. 2008 May;19(3):235-44. doi: 10.1097/FBP.0b013e3282feb0c1. Behav Pharmacol. 2008. PMID: 18469541
-
A novel dualistic profile of an allosteric AMPA receptor modulator identified through studies on recombinant receptors, mouse hippocampal synapses and crystal structures.Neuroscience. 2015 Dec 3;310:709-22. doi: 10.1016/j.neuroscience.2015.09.073. Epub 2015 Oct 9. Neuroscience. 2015. PMID: 26450748
-
AMPA receptor potentiators for the treatment of CNS disorders.Curr Drug Targets CNS Neurol Disord. 2004 Jun;3(3):181-94. doi: 10.2174/1568007043337508. Curr Drug Targets CNS Neurol Disord. 2004. PMID: 15180479 Review.
-
S18986: a positive modulator of AMPA-receptors enhances (S)-AMPA-mediated BDNF mRNA and protein expression in rat primary cortical neuronal cultures.Eur J Pharmacol. 2007 Apr 30;561(1-3):23-31. doi: 10.1016/j.ejphar.2007.01.030. Epub 2007 Feb 1. Eur J Pharmacol. 2007. PMID: 17331496
Cited by
-
Synergistic enhancing-memory effect of donepezil and S 47445, an AMPA positive allosteric modulator, in middle-aged and aged mice.Psychopharmacology (Berl). 2018 Mar;235(3):771-787. doi: 10.1007/s00213-017-4792-5. Epub 2017 Nov 22. Psychopharmacology (Berl). 2018. PMID: 29167913 Free PMC article.
-
The likelihood of cognitive enhancement.Pharmacol Biochem Behav. 2011 Aug;99(2):116-29. doi: 10.1016/j.pbb.2010.12.024. Epub 2011 Jan 6. Pharmacol Biochem Behav. 2011. PMID: 21215768 Free PMC article. Review.
-
New Insights into Alzheimer's Disease: Novel Pathogenesis, Drug Target and Delivery.Pharmaceutics. 2023 Apr 3;15(4):1133. doi: 10.3390/pharmaceutics15041133. Pharmaceutics. 2023. PMID: 37111618 Free PMC article. Review.
-
Positive AMPA and Kainate Receptor Modulators and Their Therapeutic Potential in CNS Diseases: A Comprehensive Review.Int J Mol Sci. 2025 Jul 4;26(13):6450. doi: 10.3390/ijms26136450. Int J Mol Sci. 2025. PMID: 40650226 Free PMC article. Review.
-
Strategies for Treatment of Disease-Associated Dementia Beyond Alzheimer's Disease: An Update.Curr Neuropharmacol. 2023;21(2):309-339. doi: 10.2174/1570159X20666220411083922. Curr Neuropharmacol. 2023. PMID: 35410602 Free PMC article. Review.
References
-
- Lynch G. Glutamate‐based therapeutic approaches: Ampakines. Curr Opin Pharmacol 2006;6:82–88. - PubMed
-
- Morrow JA, Maclean JKF, Jamieson C. Recent advances in positive allosteric modulators of the AMPA receptor. Curr Opin Drug Discov Devel 2006;9:571–579. - PubMed
-
- Advokat C, Pellegrin AI. Excitatory amino acids and memory: Evidence from research on Alzheimer's disease and behavioral pharmacology. Spring 1992;16:13–24. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

