Chemokine receptor CXCR3 promotes growth of glioma
- PMID: 21051441
- PMCID: PMC3026840
- DOI: 10.1093/carcin/bgq224
Chemokine receptor CXCR3 promotes growth of glioma
Abstract
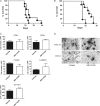
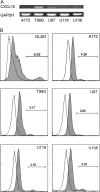
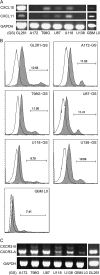
Human glioblastoma multiforme (GBM) is the most common primary brain tumor in adults. The poor prognosis and minimally successful treatments of GBM indicates a need to identify new therapeutic targets. In this study, we examined the role of CXCR3 in glioma progression using the GL261 murine model of malignant glioma. Intracranial GL261 tumors express CXCL9 and CXCL10 in vivo. Glioma-bearing CXCR3-deficient mice had significantly shorter median survival time and reduced numbers of tumor-infiltrated natural killer and natural killer T cells as compared with tumor-bearing wild-type (WT) mice. In contrast, pharmacological antagonism of CXCR3 with NBI-74330 prolonged median survival times of both tumor-bearing WT and CXCR3-deficient mice when compared with vehicle-treated groups. NBI-74330 treatment did not impact tumor infiltration of lymphocytes and microglia. A small percentage of GL261 cells were identified as CXCR3(+), which was similar to the expression of CXCR3 in several grade IV human glioma cell lines (A172, T98G, U87, U118 and U138). When cultured as gliomaspheres (GS), the human and murine lines increased CXCR3 expression; CXCR3 expression was also found in a primary human GBM-derived GS. Additionally, CXCR3 isoform A was expressed by all lines, whereas CXCR3-B was detected in T98G-, U118- and U138-GS cells. CXCL9 or CXCL10 induced in vitro glioma cell growth in GL261- and U87-GS as well as inhibited cell loss in U138-GS cells and this effect was antagonized by NBI-74330. The results suggest that CXCR3 antagonism exerts a direct anti-glioma effect and this receptor may be a potential therapeutic target for treating human GBM.
Figures



Similar articles
-
CXCL9, but not CXCL10, promotes CXCR3-dependent immune-mediated kidney disease.J Am Soc Nephrol. 2008 Jun;19(6):1177-89. doi: 10.1681/ASN.2007111179. Epub 2008 Mar 12. J Am Soc Nephrol. 2008. PMID: 18337479 Free PMC article.
-
Chemokine receptor CXCR3 and its ligands CXCL9 and CXCL10 are required for the development of murine cerebral malaria.Proc Natl Acad Sci U S A. 2008 Mar 25;105(12):4814-9. doi: 10.1073/pnas.0801544105. Epub 2008 Mar 17. Proc Natl Acad Sci U S A. 2008. PMID: 18347328 Free PMC article.
-
Immunohistochemical expression of chemokine receptor CXCR3 and its ligand CXCL10 in low-grade astrocytomas and glioblastoma multiforme: A tissue microarray-based comparison.J Cancer Res Ther. 2016 Apr-Jun;12(2):793-7. doi: 10.4103/0973-1482.153657. J Cancer Res Ther. 2016. PMID: 27461653
-
The duality of CXCR3 in glioblastoma: unveiling autocrine and paracrine mechanisms for novel therapeutic approaches.Cell Death Dis. 2023 Dec 16;14(12):835. doi: 10.1038/s41419-023-06354-2. Cell Death Dis. 2023. PMID: 38104126 Free PMC article. Review.
-
CXCR3 Ligands in Cancer and Autoimmunity, Chemoattraction of Effector T Cells, and Beyond.Front Immunol. 2020 May 29;11:976. doi: 10.3389/fimmu.2020.00976. eCollection 2020. Front Immunol. 2020. PMID: 32547545 Free PMC article. Review.
Cited by
-
CXCR3 antagonism of SDF-1(5-67) restores trabecular function and prevents retinal neurodegeneration in a rat model of ocular hypertension.PLoS One. 2012;7(6):e37873. doi: 10.1371/journal.pone.0037873. Epub 2012 Jun 4. PLoS One. 2012. PMID: 22675496 Free PMC article.
-
The Role of CXCR3 in Neurological Diseases.Curr Neuropharmacol. 2019;17(2):142-150. doi: 10.2174/1570159X15666171109161140. Curr Neuropharmacol. 2019. PMID: 29119926 Free PMC article. Review.
-
What Is Currently Known about the Role of CXCL10 in SARS-CoV-2 Infection?Int J Mol Sci. 2022 Mar 27;23(7):3673. doi: 10.3390/ijms23073673. Int J Mol Sci. 2022. PMID: 35409036 Free PMC article. Review.
-
The versatile role of HuR in Glioblastoma and its potential as a therapeutic target for a multi-pronged attack.Adv Drug Deliv Rev. 2022 Feb;181:114082. doi: 10.1016/j.addr.2021.114082. Epub 2021 Dec 16. Adv Drug Deliv Rev. 2022. PMID: 34923029 Free PMC article. Review.
-
Screening TCGA database for prognostic genes in lower grade glioma microenvironment.Ann Transl Med. 2020 Mar;8(5):209. doi: 10.21037/atm.2020.01.73. Ann Transl Med. 2020. PMID: 32309356 Free PMC article.
References
-
- Kawakami Y, et al. Dendritic cell based personalized immunotherapy based on cancer antigen research. Front. Biosci. 2008;13:1952–1958. - PubMed
-
- Vandercappellen J, et al. The role of CXC chemokines and their receptors in cancer. Cancer Lett. 2008;267:226–244. - PubMed
-
- Wendel M, et al. Natural killer cell accumulation in tumors is dependent on IFN-gamma and CXCR3 ligands. Cancer Res. 2008;68:8437–8445. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

