The influence of lamellar orientation on corneal material behavior: biomechanical and structural changes in an avian corneal disorder
- PMID: 21051696
- PMCID: PMC3101698
- DOI: 10.1167/iovs.10-5962
The influence of lamellar orientation on corneal material behavior: biomechanical and structural changes in an avian corneal disorder
Abstract
Purpose: Retinopathy, globe enlarged (RGE) is an inherited genetic disease of chickens with a corneal phenotype characterized by loss of tissue curvature and changes in peripheral collagen fibril alignment. This study aimed to characterize the material behavior of normal and RGE chicken corneas under inflation and compare this with new spatial- and depth-resolved microstructural information to investigate how stromal fibril architecture determines corneal behavior under intraocular pressure (IOP).
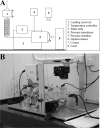
Methods: Six RGE chicken corneas and six age-matched normal controls were tested using trephinate inflation and their stress-strain behavior determined as a function of posterior pressure. Second harmonic generation mulitphoton microscopy was used to compare the in-plane appearance and degree of through-plane interlacing of collagen lamellae between normal and mutant corneas.
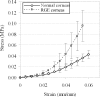
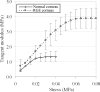
Results: RGE corneas displayed a 30-130% increase in material stiffness [E(tangent)(RGE) = 0.94 ± 0.18 MPa to 3.09 ± 0.66 MPa; E(tangent)(normals) = 0.72 ± 0.13 MPa to 1.34 ± 0.35 MPa] (P ≤ 0.05). The normal in-plane disposition of anterior collagen in the peripheral cornea was altered in RGE but through-plane lamellar interlacing was unaffected.
Conclusions: This article demonstrates changes in corneal material behavior in RGE that are qualitatively consistent with microstructural collagen alterations identified both herein and previously. This study indicates that, in general, changes in stromal fibril orientation may significantly affect corneal material behavior and thereby its response to IOP.
Figures








Similar articles
-
Ultrastructural changes in the retinopathy, globe enlarged (rge) chick cornea.J Struct Biol. 2009 May;166(2):195-204. doi: 10.1016/j.jsb.2009.01.009. Epub 2009 Mar 1. J Struct Biol. 2009. PMID: 19258040 Free PMC article.
-
Genetic, ophthalmic, morphometric and histopathological analysis of the Retinopathy Globe Enlarged (rge) chicken.Mol Vis. 2003 Jul 1;9:295-300. Mol Vis. 2003. PMID: 12847422
-
Collagen organization in the chicken cornea and structural alterations in the retinopathy, globe enlarged (rge) phenotype--an X-ray diffraction study.J Struct Biol. 2008 Jan;161(1):1-8. doi: 10.1016/j.jsb.2007.08.015. Epub 2007 Sep 7. J Struct Biol. 2008. PMID: 17936639
-
The use of X-ray scattering techniques to quantify the orientation and distribution of collagen in the corneal stroma.Prog Retin Eye Res. 2009 Sep;28(5):369-92. doi: 10.1016/j.preteyeres.2009.06.005. Epub 2009 Jul 3. Prog Retin Eye Res. 2009. PMID: 19577657 Review.
-
Evolution of the vertebrate corneal stroma.Prog Retin Eye Res. 2018 May;64:65-76. doi: 10.1016/j.preteyeres.2018.01.002. Epub 2018 Feb 2. Prog Retin Eye Res. 2018. PMID: 29398348 Review.
Cited by
-
Live human assessment of depth-dependent corneal displacements with swept-source optical coherence elastography.PLoS One. 2018 Dec 28;13(12):e0209480. doi: 10.1371/journal.pone.0209480. eCollection 2018. PLoS One. 2018. PMID: 30592752 Free PMC article.
-
Comparison of Patient-Specific Computational Modeling Predictions and Clinical Outcomes of LASIK for Myopia.Invest Ophthalmol Vis Sci. 2016 Nov 1;57(14):6287-6297. doi: 10.1167/iovs.16-19948. Invest Ophthalmol Vis Sci. 2016. PMID: 27893094 Free PMC article.
-
A Large-Scale Computational Analysis of Corneal Structural Response and Ectasia Risk in Myopic Laser Refractive Surgery.Trans Am Ophthalmol Soc. 2016 Aug;114:T1. Trans Am Ophthalmol Soc. 2016. PMID: 27630372 Free PMC article.
-
Comparative Corneal Histomorphometry Between Birds of Different Species.Biology (Basel). 2025 May 25;14(6):603. doi: 10.3390/biology14060603. Biology (Basel). 2025. PMID: 40563855 Free PMC article.
-
Quantitative Analysis of the Corneal Collagen Distribution after In Vivo Cross-Linking with Second Harmonic Microscopy.Biomed Res Int. 2019 Jan 10;2019:3860498. doi: 10.1155/2019/3860498. eCollection 2019. Biomed Res Int. 2019. PMID: 30756083 Free PMC article.
References
-
- Fatt I, Weissman B. Physiology of the Eye: An Introduction to the Vegetative Functions. 2nd ed. Boston: Butterworth-Heinmann; 1992
-
- Meek KM. The cornea and sclera. In: Fratzl P, ed. Collagen: Structure and Mechanics. New York: Springer; 2008: 359–396
-
- Boote C, Dennis S, Huang Y, Meek K. Lamellar orientation in human cornea in relation to mechanical properties. J Struct Biol. 2005;149:1–6 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

