Viable mouse gene ablations that robustly alter brain Aβ levels are rare
- PMID: 21054826
- PMCID: PMC2988800
- DOI: 10.1186/1471-2202-11-143
Viable mouse gene ablations that robustly alter brain Aβ levels are rare
Abstract
Background: Accumulation of amyloid-β (Aβ) peptide in the brain is thought to play a key pathological role in Alzheimer's disease. Many pharmacological targets have therefore been proposed based upon the biochemistry of Aβ, but not all are equally tractable for drug discovery.
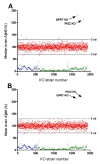
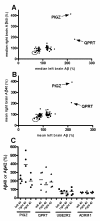
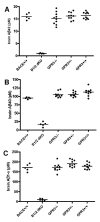
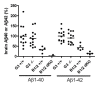
Results: To search for novel targets that affect brain Aβ without causing toxicity, we screened mouse brain samples from 1930 novel gene knock-out (KO) strains, representing 1926 genes, using Aβ ELISA assays. Although robust Aβ lowering was readily apparent in brains from a BACE1 KO strain, none of the novel strains exhibited robust decreases in brain Aβ, including a GPR3 KO strain, which had previously been proposed as an Aβ target. However, significantly increased Aβ was observed in brain samples from two KO strains, corresponding to genes encoding the glycosylphosphatidylinositol mannosyl transferase PIGZ and quinolinate phosphoribosyltransferase (QPRT).
Conclusions: Thus, gene ablations that are permissive for mouse survival and that also have a robust effect on Aβ levels in the brain are rare.
Figures

Similar articles
-
Effects of BACE1 haploinsufficiency on APP processing and Aβ concentrations in male and female 5XFAD Alzheimer mice at different disease stages.Neuroscience. 2015 Oct 29;307:128-37. doi: 10.1016/j.neuroscience.2015.08.037. Epub 2015 Aug 24. Neuroscience. 2015. PMID: 26314636 Free PMC article.
-
Alternative Selection of β-Site APP-Cleaving Enzyme 1 (BACE1) Cleavage Sites in Amyloid β-Protein Precursor (APP) Harboring Protective and Pathogenic Mutations within the Aβ Sequence.J Biol Chem. 2016 Nov 11;291(46):24041-24053. doi: 10.1074/jbc.M116.744722. Epub 2016 Sep 29. J Biol Chem. 2016. PMID: 27687728 Free PMC article.
-
Presynaptic dystrophic neurites surrounding amyloid plaques are sites of microtubule disruption, BACE1 elevation, and increased Aβ generation in Alzheimer's disease.Acta Neuropathol. 2016 Aug;132(2):235-256. doi: 10.1007/s00401-016-1558-9. Epub 2016 Mar 18. Acta Neuropathol. 2016. PMID: 26993139 Free PMC article.
-
The beta-secretase, BACE: a prime drug target for Alzheimer's disease.J Mol Neurosci. 2001 Oct;17(2):157-70. doi: 10.1385/JMN:17:2:157. J Mol Neurosci. 2001. PMID: 11816789 Review.
-
BACE1: the beta-secretase enzyme in Alzheimer's disease.J Mol Neurosci. 2004;23(1-2):105-14. doi: 10.1385/JMN:23:1-2:105. J Mol Neurosci. 2004. PMID: 15126696 Review.
Cited by
-
Identification and Preclinical Pharmacology of the γ-Secretase Modulator BMS-869780.Int J Alzheimers Dis. 2014;2014:431858. doi: 10.1155/2014/431858. Epub 2014 Jul 8. Int J Alzheimers Dis. 2014. PMID: 25097793 Free PMC article.
-
Crystal structures of human 3-hydroxyanthranilate 3,4-dioxygenase with native and non-native metals bound in the active site.Acta Crystallogr D Struct Biol. 2017 Apr 1;73(Pt 4):340-348. doi: 10.1107/S2059798317002029. Epub 2017 Mar 31. Acta Crystallogr D Struct Biol. 2017. PMID: 28375145 Free PMC article.
-
Dynamics of endogenous Hsp70 synthesis in the brain of olfactory bulbectomized mice.Cell Stress Chaperones. 2013 Jan;18(1):109-18. doi: 10.1007/s12192-012-0359-x. Epub 2012 Jul 27. Cell Stress Chaperones. 2013. PMID: 22836235 Free PMC article.
-
Effects of sub-chronic donepezil on brain Abeta and cognition in a mouse model of Alzheimer's disease.Psychopharmacology (Berl). 2013 Nov;230(2):279-89. doi: 10.1007/s00213-013-3152-3. Epub 2013 Jun 20. Psychopharmacology (Berl). 2013. PMID: 23783773
-
Predicting human disease mutations and identifying drug targets from mouse gene knockout phenotyping campaigns.Dis Model Mech. 2019 May 7;12(5):dmm038224. doi: 10.1242/dmm.038224. Dis Model Mech. 2019. PMID: 31064765 Free PMC article. Review.
References
-
- Sambamurti K, Hardy J, Refolo LM, Lahiri DK. Targeting APP metabolism for the treatment of Alzheimer's disease. Drug Dev Res. 2002;56:211–227. doi: 10.1002/ddr.10077. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

