Evaluation of the safety and efficacy of pregabalin in older patients with neuropathic pain: results from a pooled analysis of 11 clinical studies
- PMID: 21054853
- PMCID: PMC2988717
- DOI: 10.1186/1471-2296-11-85
Evaluation of the safety and efficacy of pregabalin in older patients with neuropathic pain: results from a pooled analysis of 11 clinical studies
Abstract
Background: Older patients are typically underrepresented in clinical trials of medications for chronic pain. A post hoc analysis of multiple clinical studies of pregabalin in patients with painful diabetic peripheral neuropathy (DPN) or postherpetic neuralgia (PHN) was conducted to evaluate the efficacy and safety of pregabalin in older patients.
Methods: Data from 11 double-blind, randomized, placebo-controlled clinical studies of pregabalin in patients with DPN or PHN were pooled. Efficacy outcomes included change in Daily Pain Rating Scale score, ≥30% and ≥50% responders, and endpoint pain score ≤3. Safety was based on adverse events (AEs). Primary efficacy was analyzed by analysis of covariance with terms for treatment, age category, protocol, baseline pain, and treatment-by-age category interaction.
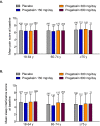
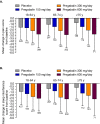
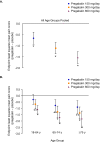
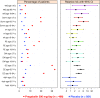
Results: 2516 patients (white, n = 2344 [93.2%]; men, n = 1347 [53.5%]; PHN, n = 1003 [39.9%]; pregabalin, n = 1595) were included in the analysis. Patients were grouped by age: 18 to 64 years (n = 1236), 65 to 74 years (n = 766), and ≥75 years (n = 514). Baseline mean pain and sleep interference scores were comparable across treatment and age groups. Significant improvements in endpoint mean pain were observed for all pregabalin dosages versus placebo in all age groups (p ≤ 0.0009), except for the lowest dosage (150 mg/day) in the youngest age group. Clinically meaningful pain relief, defined as ≥30% and ≥50% pain response, was observed in all age groups. The most common AEs were dizziness, somnolence, peripheral edema, asthenia, dry mouth, weight gain, and infections. The relative risks for these AEs increased with pregabalin dose, but did not appear related to older age or type of neuropathic pain.
Conclusions: Pregabalin (150-600 mg/day) significantly reduced pain in older patients (age ≥65 years) with neuropathic pain and improvements in pain were comparable to those observed in younger patients. Titration of pregabalin to the lowest effective dose should allow for effective pain relief while minimizing AEs in older patients with neuropathic pain. Given the common use of polypharmacy in older patients, the absence of known drug-drug interactions makes pregabalin an important treatment option for older patients with pain of neuropathic origin.
Figures
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

