K(V)4.3 N-terminal deletion mutant Δ2-39: effects on inactivation and recovery characteristics in both the absence and presence of KChIP2b
- PMID: 21057209
- PMCID: PMC3052207
- DOI: 10.4161/chan.5.1.13963
K(V)4.3 N-terminal deletion mutant Δ2-39: effects on inactivation and recovery characteristics in both the absence and presence of KChIP2b
Abstract
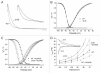
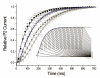
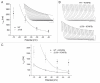
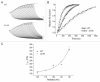
Gating transitions in the K(V)4.3 N-terminal deletion mutant Δ2-39 were characterized in the absence and presence of KChIP2b. We particularly focused on gating characteristics of macroscopic (open state) versus closed state inactivation (CSI) and recovery. In the absence of KChIP2b Δ2-39 did not significantly alter the steady-state activation "a(4)" relationship or general CSI characteristics, but it did slow the kinetics of deactivation, macroscopic inactivation, and macroscopic recovery. Recovery kinetics (for both WT K(V)4.3 and Δ2-39) were complicated and displayed sigmoidicity, a process which was enhanced by Δ2-39. Deletion of the proximal N-terminal domain therefore appeared to specifically slow mechanisms involved in regulating gating transitions occurring after the channel open state(s) had been reached. In the presence of KChIP2b Δ2-39 recovery kinetics (from both macroscopic and CSI) were accelerated, with an apparent reduction in initial sigmoidicity. Hyperpolarizing shifts in both "a(4)" and isochronal inactivation "i" were also produced. KChIP2b-mediated remodeling of K(V)4.3 gating transitions was therefore not obligatorily dependent upon an intact N-terminus. To account for these effects we propose that KChIP2 regulatory domains exist in K(V)4.3 a subunit regions outside of the proximal N-terminal. In addition to regulating macroscopic inactivation, we also propose that the K(V)4.3 N-terminus may act as a novel regulator of deactivation-recovery coupling.
Figures



Similar articles
-
The "structurally minimal" isoform KChIP2d modulates recovery of K(v)4.3 N-terminal deletion mutant Δ2-39.Channels (Austin). 2011 May-Jun;5(3):225-7. doi: 10.4161/chan.5.3.15313. Epub 2011 May 1. Channels (Austin). 2011. PMID: 21422811 Free PMC article. Review.
-
Regulation of Kv4.3 closed state inactivation and recovery by extracellular potassium and intracellular KChIP2b.Channels (Austin). 2007 Jul-Aug;1(4):305-14. doi: 10.4161/chan.5017. Epub 2007 Sep 10. Channels (Austin). 2007. PMID: 18708742
-
Regulation of Kv4.3 voltage-dependent gating kinetics by KChIP2 isoforms.J Physiol. 2004 May 15;557(Pt 1):19-41. doi: 10.1113/jphysiol.2003.058172. Epub 2004 Jan 14. J Physiol. 2004. PMID: 14724186 Free PMC article.
-
DPP10 is an inactivation modulatory protein of Kv4.3 and Kv1.4.Am J Physiol Cell Physiol. 2006 Nov;291(5):C966-76. doi: 10.1152/ajpcell.00571.2005. Epub 2006 May 31. Am J Physiol Cell Physiol. 2006. PMID: 16738002
-
Role of N-terminal domain and accessory subunits in controlling deactivation-inactivation coupling of Kv4.2 channels.Biophys J. 2008 Feb 15;94(4):1276-94. doi: 10.1529/biophysj.107.111344. Epub 2007 Nov 2. Biophys J. 2008. PMID: 17981906 Free PMC article.
Cited by
-
The "structurally minimal" isoform KChIP2d modulates recovery of K(v)4.3 N-terminal deletion mutant Δ2-39.Channels (Austin). 2011 May-Jun;5(3):225-7. doi: 10.4161/chan.5.3.15313. Epub 2011 May 1. Channels (Austin). 2011. PMID: 21422811 Free PMC article. Review.
References
-
- Birnbaum SG, Varga AW, Yuan LL, Anderson AE, Sweatt JD, Schrader LA. Structure and function of KV4-family transient potassium channels. Physiol Rev. 2004;84:803–833. - PubMed
-
- Jerng HH, Pfaffinger PJ, Covarrubias M. Molecular physiology and modulation of somatodendritic A-type potassium channels. Mol Cell Neurosci. 2004;27:343–369. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
