A multiplex assay for the simultaneous detection of antibodies against 15 Plasmodium falciparum and Anopheles gambiae saliva antigens
- PMID: 21059211
- PMCID: PMC2992071
- DOI: 10.1186/1475-2875-9-317
A multiplex assay for the simultaneous detection of antibodies against 15 Plasmodium falciparum and Anopheles gambiae saliva antigens
Abstract
Background: Assessment exposure and immunity to malaria is an important step in the fight against the disease. Increased malaria infection in non-immune travellers under anti-malarial chemoprophylaxis, as well as the implementation of malaria elimination programmes in endemic countries, raises new issues that pertain to these processes. Notably, monitoring malaria immunity has become more difficult in individuals showing low antibody (Ab) responses or taking medications against the Plasmodium falciparum blood stages. Commonly available techniques in malaria seroepidemiology have limited sensitivity, both against pre-erythrocytic, as against blood stages of the parasite. Thus, the aim of this study was to develop a sensitive tool to assess the exposure to malaria or to bites from the vector Anopheles gambiae, despite anti-malarial prophylactic treatment.
Methods: Ab responses to 13 pre-erythrocytic P. falciparum-specific peptides derived from the proteins Lsa1, Lsa3, Glurp, Salsa, Trap, Starp, CSP and Pf11.1, and to 2 peptides specific for the Anopheles gambiae saliva protein gSG6 were tested. In this study, 253 individuals from three Senegalese areas with different transmission intensities and 124 European travellers exposed to malaria during a short period of time were included.
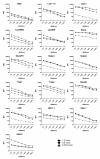
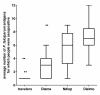
Results: The multiplex assay was optimized for most but not all of the antigens. It was rapid, reproducible and required a small volume of serum. Proportions of Ab-positive individuals, Ab levels and the mean number of antigens (Ags) recognized by each individual increased significantly with increases in the level of malaria exposure.
Conclusion: The multiplex assay developed here provides a useful tool to evaluate immune responses to multiple Ags in large populations, even when only small amounts of serum are available, or Ab titres are low, as in case of travellers. Finally, the relationship of Ab responses with malaria endemicity levels provides a way to monitor exposure in differentially exposed autochthonous individuals from various endemicity areas, as well as in travellers who are not immune, thus indirectly assessing the parasite transmission and malaria risk in the new eradication era.
Figures



References
-
- WHO. World malaria Report 2008. 2008.
-
- WHO. International travel and health. 2009.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

