Murine erythroid short-term radioprotection requires a BMP4-dependent, self-renewing population of stress erythroid progenitors
- PMID: 21060151
- PMCID: PMC2993581
- DOI: 10.1172/JCI41291
Murine erythroid short-term radioprotection requires a BMP4-dependent, self-renewing population of stress erythroid progenitors
Abstract
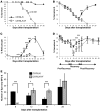
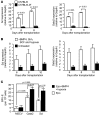
Acute anemic stress induces a systemic response designed to increase oxygen delivery to hypoxic tissues. Increased erythropoiesis is a key component of this response. Recovery from acute anemia relies on stress erythropoiesis, which is distinct from steady-state erythropoiesis. In this study we found that the bone morphogenetic protein 4-dependent (BMP4-dependent) stress erythropoiesis pathway was required and specific for erythroid short-term radioprotection following bone marrow transplantation. BMP4 signaling promoted the development of three populations of stress erythroid progenitors, which expanded in the spleen subsequent to bone marrow transplantation in mice. These progenitors did not correspond to previously identified bone marrow steady-state progenitors. The most immature population of stress progenitors was capable of self renewal while maintaining erythropoiesis without contribution to other lineages when serially transplanted into irradiated secondary and tertiary recipients. These data suggest that during the immediate post-transplant period, the microenvironment of the spleen is altered, which allows donor bone marrow cells to adopt a stress erythropoietic fate and promotes the rapid expansion and differentiation of stress erythroid progenitors. Our results also suggest that stress erythropoiesis may be manipulated through targeting the BMP4 signaling pathway to improve survival after injury.
Figures

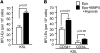


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

