Plasmodium berghei circumvents immune responses induced by merozoite surface protein 1- and apical membrane antigen 1-based vaccines
- PMID: 21060850
- PMCID: PMC2965677
- DOI: 10.1371/journal.pone.0013727
Plasmodium berghei circumvents immune responses induced by merozoite surface protein 1- and apical membrane antigen 1-based vaccines
Abstract
Background: Two current leading malaria blood-stage vaccine candidate antigens for Plasmodium falciparum, the C-terminal region of merozoite surface protein 1 (MSP1(19)) and apical membrane antigen 1 (AMA1), have been prioritized because of outstanding protective efficacies achieved in a rodent malaria Plasmodium yoelii model. However, P. falciparum vaccines based on these antigens have had disappointing outcomes in clinical trials. Discrepancies in the vaccine efficacies observed between the P. yoelii model and human clinical trials still remain problematic.
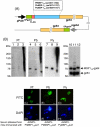
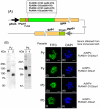
Methodology and results: In this study, we assessed the protective efficacies of a series of MSP1(19)- and AMA1-based vaccines using the P. berghei rodent malarial parasite and its transgenic models. Immunization of mice with a baculoviral-based vaccine (BBV) expressing P. falciparum MSP1(19) induced high titers of PfMSP1(19)-specific antibodies that strongly reacted with P. falciparum blood-stage parasites. However, no protection was achieved following lethal challenge with transgenic P. berghei expressing PfMSP1(19) in place of native PbMSP1(19). Similarly, neither P. berghei MSP1(19)- nor AMA1-BBV was effective against P. berghei. In contrast, immunization with P. yoelii MSP1(19)- and AMA1-BBVs provided 100% and 40% protection, respectively, against P. yoelii lethal challenge. Mice that naturally acquired sterile immunity against P. berghei became cross-resistant to P. yoelii, but not vice versa.
Conclusion: This is the first study to address blood-stage vaccine efficacies using both P. berghei and P. yoelii models at the same time. P. berghei completely circumvents immune responses induced by MSP1(19)- and AMA1-based vaccines, suggesting that P. berghei possesses additional molecules and/or mechanisms that circumvent the host's immune responses to MSP1(19) and AMA1, which are lacking in P. yoelii. Although it is not known whether P. falciparum shares these escape mechanisms with P. berghei, P. berghei and its transgenic models may have potential as useful tools for identifying and evaluating new blood-stage vaccine candidate antigens for P. falciparum.
Conflict of interest statement
Figures
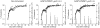
Similar articles
-
Prediction of merozoite surface protein 1 and apical membrane antigen 1 vaccine efficacies against Plasmodium chabaudi malaria based on prechallenge antibody responses.Clin Vaccine Immunol. 2009 Mar;16(3):293-302. doi: 10.1128/CVI.00230-08. Epub 2008 Dec 30. Clin Vaccine Immunol. 2009. PMID: 19116303 Free PMC article.
-
Evaluation of the safety and immunogenicity of Plasmodium falciparum apical membrane antigen 1, merozoite surface protein 1 or RTS,S vaccines with adjuvant system AS02A administered alone or concurrently in rhesus monkeys.Vaccine. 2009 Dec 11;28(2):452-62. doi: 10.1016/j.vaccine.2009.10.022. Epub 2009 Oct 24. Vaccine. 2009. PMID: 19857448
-
Merozoite surface protein 4/5 provides protection against lethal challenge with a heterologous malaria parasite strain.Infect Immun. 2004 Oct;72(10):5840-9. doi: 10.1128/IAI.72.10.5840-5849.2004. Infect Immun. 2004. PMID: 15385485 Free PMC article.
-
Plasmodium falciparum merozoite surface protein 1 as asexual blood stage malaria vaccine candidate.Expert Rev Vaccines. 2024 Jan-Dec;23(1):160-173. doi: 10.1080/14760584.2023.2295430. Epub 2023 Dec 27. Expert Rev Vaccines. 2024. PMID: 38100310 Review.
-
The carboxy-terminus of merozoite surface protein 1: structure, specific antibodies and immunity to malaria.Parasitology. 2009 Oct;136(12):1445-56. doi: 10.1017/S0031182009990515. Epub 2009 Jul 23. Parasitology. 2009. PMID: 19627632 Review.
Cited by
-
Protective efficacy of baculovirus dual expression system vaccine expressing Plasmodium falciparum circumsporozoite protein.PLoS One. 2013 Aug 12;8(8):e70819. doi: 10.1371/journal.pone.0070819. eCollection 2013. PLoS One. 2013. PMID: 23951015 Free PMC article.
-
Eimeria maxima: efficacy of recombinant Mycobacterium bovis BCG expressing apical membrane antigen1 against homologous infection.Parasitol Res. 2013 Nov;112(11):3825-33. doi: 10.1007/s00436-013-3570-5. Epub 2013 Aug 15. Parasitol Res. 2013. PMID: 23949244
-
Baculovirus-vectored multistage Plasmodium vivax vaccine induces both protective and transmission-blocking immunities against transgenic rodent malaria parasites.Infect Immun. 2014 Oct;82(10):4348-57. doi: 10.1128/IAI.02040-14. Epub 2014 Aug 4. Infect Immun. 2014. PMID: 25092912 Free PMC article.
-
Baculovirus Display of Peptides and Proteins for Medical Applications.Viruses. 2023 Feb 1;15(2):411. doi: 10.3390/v15020411. Viruses. 2023. PMID: 36851625 Free PMC article. Review.
-
Protective Immunity in Mice Immunized With P. vivax MSP119-Based Formulations and Challenged With P. berghei Expressing PvMSP119.Front Immunol. 2020 Feb 19;11:28. doi: 10.3389/fimmu.2020.00028. eCollection 2020. Front Immunol. 2020. PMID: 32153555 Free PMC article.
References
-
- Kocken CH, Withers-Martinez C, Dubbeld MA, van der Wel A, Hackett F, et al. High-level expression of the malaria blood-stage vaccine candidate Plasmodium falciparum apical membrane antigen 1 and induction of antibodies that inhibit erythrocyte invasion. Infect Immun. 2002;70:4471–4476. - PMC - PubMed
-
- Holder AA. The precursor to major merozoite surface antigens: structure and role in immunity. Prog Allergy. 1988;41:72–97. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

