Critical role for IL-21 in both primary and memory anti-viral CD8+ T-cell responses
- PMID: 21061439
- PMCID: PMC3119210
- DOI: 10.1002/eji.200939939
Critical role for IL-21 in both primary and memory anti-viral CD8+ T-cell responses
Abstract
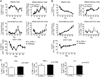
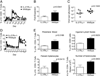
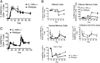
While it is well established that CD8(+) T cells generated in the absence of CD4(+) T cells mediate defective recall responses, the mechanism by which CD4(+) T cells confer help in the generation of CD8(+) T-cell responses remains poorly understood. To determine whether CD4(+) T-cell-derived IL-21 is an important regulator of CD8(+) T-cell responses in help-dependent and -independent viral infections, we examined these responses in the IL-21Rα(-/-) mouse model. We show that IL-21 has a role in primary CD8(+) T-cell responses and in recall CD8(+) T-cell responses in help-dependent viral infections. This effect is due to a direct action of IL-21 in enhancing the proliferation of virus-specific CD8(+) T cells and reducing their TRAIL expression. These findings indicate that IL-21 is an important mediator of CD4(+) T-cell help to CD8(+) T cells.
Copyright © 2010 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Conflict of interest statement
Figures



References
-
- Janssen EM, Droin NM, Lemmens EE, Pinkoski MJ, Bensinger SJ. CD4+T-cell help controls CD8+T-cell memory via TRAIL-mediated activation-induced cell death. Nature. 2005;434:88. - PubMed
-
- Bevan MJ. Helping the CD8(+) T-cell response. Nat. Rev. Immunol. 2004;4:595–602. - PubMed
-
- Livingstone AM, Wilson EB, Ontiveros F, Wang JC. Unravelling the mechanisms of help for CD8+T cell responses. Immunol. Res. 2009;45:209–217. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

