Dopaminergic regulation of sleep and cataplexy in a murine model of narcolepsy
- PMID: 21061851
- PMCID: PMC2941415
- DOI: 10.1093/sleep/33.10.1295
Dopaminergic regulation of sleep and cataplexy in a murine model of narcolepsy
Abstract
Study objectives: To determine if the dopaminergic system modulates cataplexy, sleep attacks and sleep-wake behavior in narcoleptic mice.
Design: Hypocretin/orexin knockout (i.e., narcoleptic) and wild-type mice were administered amphetamine and specific dopamine receptor modulators to determine their effects on sleep, cataplexy and sleep attacks.
Patients or participants: Hypocretin knockout (n = 17) and wild-type mice (n = 21).
Interventions: Cataplexy, sleep attacks and sleep-wake behavior were identified using electroencephalogram, electromyogram and videography. These behaviors were monitored for 4 hours after an i.p. injection of saline, amphetamine and specific dopamine receptor modulators (D1- and D2-like receptor modulators).
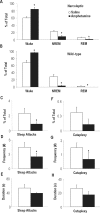
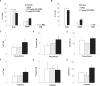
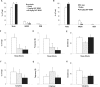
Measurements and results: Amphetamine (2 mg/kg), which increases brain dopamine levels, decreased sleep attacks and cataplexy by 61% and 67%, suggesting that dopamine transmission modulates such behaviors. Dopamine receptor modulation also had powerful effects on sleep attacks and cataplexy. Activation (SKF 38393; 20 mg/kg) and blockade (SCH 23390; 1 mg/kg) of D1-like receptors decreased and increased sleep attacks by 77% and 88%, without affecting cataplexy. Pharmacological activation of D2-like receptors (quinpirole; 0.5 mg/kg) increased cataplectic attacks by 172% and blockade of these receptors (eticlopride; 1 mg/kg) potently suppressed them by 97%. Manipulation of D2-like receptors did not affect sleep attacks.
Conclusions: We show that the dopaminergic system plays a role in regulating both cataplexy and sleep attacks in narcoleptic mice. We found that cataplexy is modulated by a D2-like receptor mechanism, whereas dopamine modulates sleep attacks by a D1-like receptor mechanism. These results support a role for the dopamine system in regulating sleep attacks and cataplexy in a murine model of narcolepsy.
Figures



Similar articles
-
Local administration of dopaminergic drugs into the ventral tegmental area modulates cataplexy in the narcoleptic canine.Brain Res. 1996 Sep 9;733(1):83-100. doi: 10.1016/0006-8993(96)00541-0. Brain Res. 1996. PMID: 8891251 Free PMC article.
-
Dopamine D3 agonists into the substantia nigra aggravate cataplexy but do not modify sleep [corrected].Neuroreport. 1999 Sep 29;10(14):3111-8. doi: 10.1097/00001756-199909290-00043. Neuroreport. 1999. Corrected and republished in: Neuroreport. 1999 Nov 26;10(17):3717-24. doi: 10.1097/00001756-199911260-00046. PMID: 10549832 Corrected and republished.
-
Effects of dopaminergic drugs on innate pheromone-mediated reward in female mice: a new case of dopamine-independent "liking.".Behav Neurosci. 2007 Oct;121(5):920-32. doi: 10.1037/0735-7044.121.5.920. Behav Neurosci. 2007. PMID: 17907824
-
A consensus definition of cataplexy in mouse models of narcolepsy.Sleep. 2009 Jan;32(1):111-6. Sleep. 2009. PMID: 19189786 Free PMC article.
-
Rewiring brain circuits to block cataplexy in murine models of narcolepsy.Curr Opin Neurobiol. 2017 Jun;44:110-115. doi: 10.1016/j.conb.2017.03.017. Epub 2017 Apr 23. Curr Opin Neurobiol. 2017. PMID: 28445807 Free PMC article. Review.
Cited by
-
VTA dopaminergic neurons regulate ethologically relevant sleep-wake behaviors.Nat Neurosci. 2016 Oct;19(10):1356-66. doi: 10.1038/nn.4377. Epub 2016 Sep 5. Nat Neurosci. 2016. PMID: 27595385 Free PMC article.
-
Impaired GABA and glycine transmission triggers cardinal features of rapid eye movement sleep behavior disorder in mice.J Neurosci. 2011 May 11;31(19):7111-21. doi: 10.1523/JNEUROSCI.0347-11.2011. J Neurosci. 2011. PMID: 21562273 Free PMC article.
-
Dopamine D1 and D2 receptors mediate analgesic and hypnotic effects of l-tetrahydropalmatine in a mouse neuropathic pain model.Psychopharmacology (Berl). 2019 Nov;236(11):3169-3182. doi: 10.1007/s00213-019-05275-3. Epub 2019 Jun 6. Psychopharmacology (Berl). 2019. PMID: 31172225
-
Complex movement disorders at disease onset in childhood narcolepsy with cataplexy.Brain. 2011 Dec;134(Pt 12):3477-89. doi: 10.1093/brain/awr244. Epub 2011 Sep 19. Brain. 2011. PMID: 21930661 Free PMC article.
-
Cataplexy--clinical aspects, pathophysiology and management strategy.Nat Rev Neurol. 2014 Jul;10(7):386-95. doi: 10.1038/nrneurol.2014.97. Epub 2014 Jun 3. Nat Rev Neurol. 2014. PMID: 24890646 Free PMC article. Review.
References
-
- Peyron C, Faraco J, Rogers W, et al. A mutation in a case of early onset narcolepsy and a generalized absence of hypocretin peptides in human narcoleptic brains. Nat Med. 2000;6:991–7. - PubMed
-
- Eisensehr I, Linke R, Tatsch K, et al. Alteration of the striatal dopaminergic system in human narcolepsy. Neurology. 2003;60:1817–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

