Absolute configuration of (-)-gambogic acid, an antitumor agent
- PMID: 21067206
- PMCID: PMC3052414
- DOI: 10.1021/np100422z
Absolute configuration of (-)-gambogic acid, an antitumor agent
Abstract
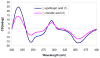
(-)-Gambogic acid (1), a biologically active "caged xanthone" from gamboge, the dried resin of Garcinia hanburyi, is of interest as a potential anticancer agent. The planar structure of (-)-gambogic acid has been determined previously by analysis of its detailed NMR data and confirmed by single-crystal X-ray diffraction, with the absolute configuration at C-13 deduced as R through a series of chemical degradations. Using (-)-morellic acid (2), an analogue of (-)-gambogic acid, as a model compound, the 5R, 7S, 10aS, 13R, 27S absolute configuration of (-)-gambogic acid was determined for the first time by comparison of physical and spectroscopic data, especially experimental and calculated electronic circular dichroism.
Figures
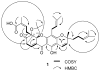
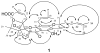
Similar articles
-
Cytotoxic polyprenylated xanthones from the resin of Garcinia hanburyi.J Nat Prod. 2009 Jan;72(1):117-24. doi: 10.1021/np800460b. J Nat Prod. 2009. PMID: 19072548
-
Three new xanthones from the resin of Garcinia hanburyi.Planta Med. 2013 Jun;79(9):792-6. doi: 10.1055/s-0032-1328537. Epub 2013 May 13. Planta Med. 2013. PMID: 23670620
-
A new xanthone derivative from twigs of Garcinia nobilis.Nat Prod Res. 2014;28(14):1030-6. doi: 10.1080/14786419.2014.903398. Epub 2014 Apr 4. Nat Prod Res. 2014. PMID: 24697710
-
[Recent advances in chemistry and biology of gamboge].Zhongguo Zhong Yao Za Zhi. 2013 Jan;38(1):19-25. Zhongguo Zhong Yao Za Zhi. 2013. PMID: 23596869 Review. Chinese.
-
[Progress in research of the structural optimization of natural product-like Garcinia caged xanthones].Yao Xue Xue Bao. 2014 Mar;49(3):293-302. Yao Xue Xue Bao. 2014. PMID: 24961098 Review. Chinese.
Cited by
-
Gambogic acid protects LPS-induced apoptosis and inflammation in a cell model of neonatal pneumonia through the regulation of TrkA/Akt signaling pathway.BMC Pharmacol Toxicol. 2021 May 11;22(1):28. doi: 10.1186/s40360-021-00496-9. BMC Pharmacol Toxicol. 2021. PMID: 33971977 Free PMC article.
-
Molecular targets of gambogic acid in cancer: recent trends and advancements.Tumour Biol. 2016 Oct;37(10):12915-12925. doi: 10.1007/s13277-016-5194-8. Epub 2016 Jul 22. Tumour Biol. 2016. PMID: 27448303 Review.
-
Anticancer Inhibitors of Hsp90 Function: Beyond the Usual Suspects.Adv Cancer Res. 2016;129:51-88. doi: 10.1016/bs.acr.2015.12.001. Epub 2016 Feb 10. Adv Cancer Res. 2016. PMID: 26916001 Free PMC article. Review.
-
Gambogic acid, a natural product inhibitor of Hsp90.J Nat Prod. 2011 May 27;74(5):1085-92. doi: 10.1021/np200029q. Epub 2011 Apr 12. J Nat Prod. 2011. PMID: 21486005 Free PMC article.
-
Caged Polyprenylated Xanthones in Garcinia hanburyi and the Biological Activities of Them.Drug Des Devel Ther. 2023 Dec 5;17:3625-3660. doi: 10.2147/DDDT.S426685. eCollection 2023. Drug Des Devel Ther. 2023. PMID: 38076632 Free PMC article. Review.
References
-
- Asano J, Chiba K, Tada M, Yoshii T. Phytochemistry. 1996;41:815–820. - PubMed
-
- Han QB, Wang YL, Yang L, Tso TF, Qiao CF, Song JZ, Xu LJ, Chen SL, Yang DJ, Xu HX. Chem Pharm Bull. 2006;54:265–267. - PubMed
-
- Reutrakul V, Anantachoke N, Pohmakotr M, Jaipetch T, Sophasan S, Yoosook C, Kasisit J, Napaswat C, Santisuk T, Tuchinda P. Planta Med. 2007;73:33–40. - PubMed
-
- Tao SJ, Guan SH, Wang W, Lu ZQ, Chen GT, Sha N, Yue QX, Liu X, Guo DA. J Nat Prod. 2009;72:117–124. - PubMed
-
- Han Q-B, Xu H-X. Curr Med Chem. 2009;16:3775–3796. It is stated on page 3781 of this comprehensive review that the absolute configuration of (−)-gambogic acid and its analogues remained undetermined at the time of publication. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

