Stem-cell gene therapy for the Wiskott-Aldrich syndrome
- PMID: 21067383
- PMCID: PMC3064520
- DOI: 10.1056/NEJMoa1003548
Stem-cell gene therapy for the Wiskott-Aldrich syndrome
Abstract
The Wiskott-Aldrich syndrome (WAS) is an X-linked recessive primary immunodeficiency disorder associated with thrombocytopenia, eczema, and autoimmunity. We treated two patients who had this disorder with a transfusion of autologous, genetically modified hematopoietic stem cells (HSC). We found sustained expression of WAS protein expression in HSC, lymphoid and myeloid cells, and platelets after gene therapy. T and B cells, natural killer (NK) cells, and monocytes were functionally corrected. After treatment, the patients' clinical condition markedly improved, with resolution of hemorrhagic diathesis, eczema, autoimmunity, and predisposition to severe infection. Comprehensive insertion-site analysis showed vector integration that targeted multiple genes controlling growth and immunologic responses in a persistently polyclonal hematopoiesis. (Funded by Deutsche Forschungsgemeinschaft and others; German Clinical Trials Register number, DRKS00000330.).
Figures
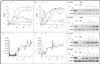


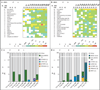
References
-
- Puck JM, Candotti F. Lessons from the Wiskott–Aldrich syndrome. N Engl J Med. 2006;355:1759–1761. - PubMed
-
- Notarangelo LD, Miao CH, Ochs HD. Wiskott-Aldrich syndrome. Curr Opin Hematol. 2008;15:30–36. - PubMed
-
- Derry JM, Ochs HD, Francke U. Isolation of a novel gene mutated in Wiskott-Aldrich syndrome. Cell. 1994;78:635–644. - PubMed
-
- Thrasher AJ, Burns SO. WASP: a key immunological multitasker. Nat Rev Immunol. 2010;10:182–192. - PubMed
-
- Bosticardo M, Marangoni F, Aiuti A, Villa A, Grazia Roncarolo M. Recent advances in understanding the pathophysiology of Wiskott-Aldrich syndrome. Blood. 2009;113:6288–6295. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
