Early life stress enhances behavioral vulnerability to stress through the activation of REST4-mediated gene transcription in the medial prefrontal cortex of rodents
- PMID: 21068306
- PMCID: PMC6633839
- DOI: 10.1523/JNEUROSCI.1436-10.2010
Early life stress enhances behavioral vulnerability to stress through the activation of REST4-mediated gene transcription in the medial prefrontal cortex of rodents
Abstract
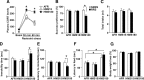
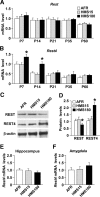
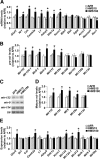
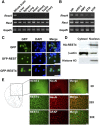
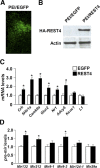
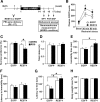
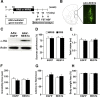
There is growing evidence suggesting that early life events have long-term effects on the neuroendocrine and behavioral developments of rodents. However, little is known about the involvement of early life events in the susceptibility to subsequent stress exposure during adulthood. The present study characterized the effect of maternal separation, an animal model of early life adversity, on the behavioral response to repeated restraint stress in adult rats and investigated the molecular mechanism underlying behavioral vulnerability to chronic stress induced by the maternal separation. Rat pups were separated from the dams for 180 min per day from postnatal day 2 through 14 (HMS180 rats). We found that, as young adults, HMS180 rats showed a greater hypothalamic-pituitary-adrenal axis response to acute restraint stress than nonseparated control rats. In addition, repeatedly restrained HMS180 rats showed increased depression-like behavior and an anhedonic response compared with nonrestrained HMS180 rats. Furthermore, HMS180 rats showed increased expression of REST4, a neuron-specific splicing variant of the transcriptional repressor REST (repressor element-1 silencing transcription factor), and a variety of REST target gene mRNAs and microRNAs in the medial prefrontal cortex (mPFC). Finally, REST4 overexpression in the mPFC of neonatal mice via polyethyleneimine-mediated gene transfer enhanced the expression of its target genes as well as behavioral vulnerability to repeated restraint stress. In contrast, REST4 overexpression in the mPFC of adult mice did not affect depression-like behaviors after repeated stress exposure. These results suggest that the activation of REST4-mediated gene regulation in the mPFC during postnatal development is involved in stress vulnerability.
Figures
Similar articles
-
Differential neuroendocrine responses to chronic variable stress in adult Long Evans rats exposed to handling-maternal separation as neonates.Psychoneuroendocrinology. 2005 Jul;30(6):520-33. doi: 10.1016/j.psyneuen.2004.12.004. Psychoneuroendocrinology. 2005. PMID: 15808921
-
Effect of repeated restraint on homotypic stress-induced nitric oxide synthases expression in brain structures regulating HPA axis.Pharmacol Rep. 2012;64(6):1381-90. doi: 10.1016/s1734-1140(12)70935-0. Pharmacol Rep. 2012. PMID: 23406748
-
An unexpected increase in restraint duration alters the expression of stress response habituation.Physiol Behav. 2013 Oct 2;122:193-200. doi: 10.1016/j.physbeh.2013.03.029. Epub 2013 Apr 6. Physiol Behav. 2013. PMID: 23566924 Free PMC article.
-
Epigenetic programming of the stress response in male and female rats by prenatal restraint stress.Brain Res Rev. 2008 Mar;57(2):571-85. doi: 10.1016/j.brainresrev.2007.11.004. Epub 2007 Nov 28. Brain Res Rev. 2008. PMID: 18164765 Review.
-
Oxidative and nitrosative stress pathways in the brain of socially isolated adult male rats demonstrating depressive- and anxiety-like symptoms.Brain Struct Funct. 2017 Jan;222(1):1-20. doi: 10.1007/s00429-016-1218-9. Epub 2016 Mar 31. Brain Struct Funct. 2017. PMID: 27033097 Review.
Cited by
-
Hippocampal MicroRNA-124 Enhances Chronic Stress Resilience in Mice.J Neurosci. 2016 Jul 6;36(27):7253-67. doi: 10.1523/JNEUROSCI.0319-16.2016. J Neurosci. 2016. PMID: 27383599 Free PMC article.
-
Age‑related differences for expression of the nerve‑specific proteins after peripheral nerve injury.Exp Ther Med. 2022 Sep 21;24(5):682. doi: 10.3892/etm.2022.11618. eCollection 2022 Nov. Exp Ther Med. 2022. PMID: 36185767 Free PMC article.
-
Early-life adversity and neurological disease: age-old questions and novel answers.Nat Rev Neurol. 2019 Nov;15(11):657-669. doi: 10.1038/s41582-019-0246-5. Epub 2019 Sep 17. Nat Rev Neurol. 2019. PMID: 31530940 Free PMC article. Review.
-
The effects of early life stress on reward processing.J Psychiatr Res. 2018 Jun;101:80-103. doi: 10.1016/j.jpsychires.2018.02.002. Epub 2018 Feb 13. J Psychiatr Res. 2018. PMID: 29567510 Free PMC article. Review.
-
A mechanistic look at the effects of adversity early in life on cardiovascular disease risk during adulthood.Acta Physiol (Oxf). 2014 Feb;210(2):277-87. doi: 10.1111/apha.12189. Epub 2013 Dec 13. Acta Physiol (Oxf). 2014. PMID: 24330084 Free PMC article. Review.
References
-
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. Ed 4. Washington, DC: American Psychiatric Association; 1994.
-
- Armario A, Martí O, Vallès A, Dal-Zotto S, Ons S. Long-term effects of a single exposure to immobilization on the hypothalamic-pituitary-adrenal axis: neurobiologic mechanisms. Ann N Y Acad Sci. 2004;1018:162–172. - PubMed
-
- Ballas N, Battaglioli E, Atouf F, Andres ME, Chenoweth J, Anderson ME, Burger C, Moniwa M, Davie JR, Bowers WJ, Federoff HJ, Rose DW, Rosenfeld MG, Brehm P, Mandel G. Regulation of neuronal traits by a novel transcriptional complex. Neuron. 2001;31:353–365. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
