The major alpha-tubulin K40 acetyltransferase alphaTAT1 promotes rapid ciliogenesis and efficient mechanosensation
- PMID: 21068373
- PMCID: PMC3003046
- DOI: 10.1073/pnas.1013728107
The major alpha-tubulin K40 acetyltransferase alphaTAT1 promotes rapid ciliogenesis and efficient mechanosensation
Abstract
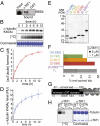
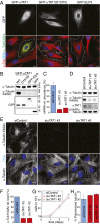
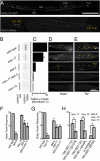
Long-lived microtubules found in ciliary axonemes, neuronal processes, and migrating cells are marked by α-tubulin acetylation on lysine 40, a modification that takes place inside the microtubule lumen. The physiological importance of microtubule acetylation remains elusive. Here, we identify a BBSome-associated protein that we name αTAT1, with a highly specific α-tubulin K40 acetyltransferase activity and a catalytic preference for microtubules over free tubulin. In mammalian cells, the catalytic activity of αTAT1 is necessary and sufficient for α-tubulin K40 acetylation. Remarkably, αTAT1 is universally and exclusively conserved in ciliated organisms, and is required for the acetylation of axonemal microtubules and for the normal kinetics of primary cilium assembly. In Caenorhabditis elegans, microtubule acetylation is most prominent in touch receptor neurons (TRNs) and MEC-17, a homolog of αTAT1, and its paralog αTAT-2 are required for α-tubulin acetylation and for two distinct types of touch sensation. Furthermore, in animals lacking MEC-17, αTAT-2, and the sole C. elegans K40α-tubulin MEC-12, touch sensation can be restored by expression of an acetyl-mimic MEC-12[K40Q]. We conclude that αTAT1 is the major and possibly the sole α-tubulin K40 acetyltransferase in mammals and nematodes, and that tubulin acetylation plays a conserved role in several microtubule-based processes.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Comment in
-
Tubulin acetyltransferase discovered: ciliary role in the ancestral eukaryote expanded to neurons in metazoans.Proc Natl Acad Sci U S A. 2010 Dec 14;107(50):21238-9. doi: 10.1073/pnas.1016396108. Epub 2010 Dec 6. Proc Natl Acad Sci U S A. 2010. PMID: 21135216 Free PMC article. No abstract available.
References
-
- Nogales E, Wolf SG, Downing KH. Structure of the alpha beta tubulin dimer by electron crystallography. Nature. 1998;391:199–203. - PubMed
-
- Kozminski KG, Diener DR, Rosenbaum JL. High level expression of nonacetylatable alpha-tubulin in Chlamydomonas reinhardtii. Cell Motil Cytoskeleton. 1993;25:158–170. - PubMed
-
- Fukushige T, et al. MEC-12, an alpha-tubulin required for touch sensitivity in C. elegans. J Cell Sci. 1999;112:395–403. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

