Genetic defects of GDF6 in the zebrafish out of sight mutant and in human eye developmental anomalies
- PMID: 21070663
- PMCID: PMC2992036
- DOI: 10.1186/1471-2156-11-102
Genetic defects of GDF6 in the zebrafish out of sight mutant and in human eye developmental anomalies
Abstract
Background: The size of the vertebrate eye and the retina is likely to be controlled at several stages of embryogenesis by mechanisms that affect cell cycle length as well as cell survival. A mutation in the zebrafish out of sight (out) locus results in a particularly severe reduction of eye size. The goal of this study is to characterize the outm233 mutant, and to determine whether mutations in the out gene cause microphthalmia in humans.
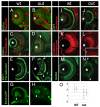
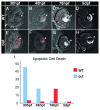
Results: In this study, we show that the severe reduction of eye size in the outm233 mutant is caused by a mutation in the zebrafish gdf6a gene. Despite the small eye size, the overall retinal architecture appears largely intact, and immunohistochemical studies confirm that all major cell types are present in outm233 retinae. Subtle cell fate and patterning changes are present predominantly in amacrine interneurons. Acridine orange and TUNEL staining reveal that the levels of apoptosis are abnormally high in outm233 mutant eyes during early neurogenesis. Mutation analysis of the GDF6 gene in 200 patients with microphthalmia revealed amino acid substitutions in four of them. In two patients additional skeletal defects were observed.
Conclusions: This study confirms the essential role of GDF6 in the regulation of vertebrate eye size. The reduced eye size in the zebrafish outm233 mutant is likely to be caused by a transient wave of apoptosis at the onset of neurogenesis. Amino acid substitutions in GDF6 were detected in 4 (2%) of 200 patients with microphthalmia. In two patients different skeletal defects were also observed, suggesting pleitrophic effects of GDF6 variants. Parents carrying these variants are asymptomatic, suggesting that GDF6 sequence alterations are likely to contribute to the phenotype, but are not the sole cause of the disease. Variable expressivity and penetrance suggest a complex non-Mendelian inheritance pattern where other genetic factors may influence the outcome of the phenotype.
Figures





Similar articles
-
Apoptotic and proliferative defects characterize ocular development in a microphthalmic BMP model.Invest Ophthalmol Vis Sci. 2013 Jul 10;54(7):4636-47. doi: 10.1167/iovs.13-11674. Invest Ophthalmol Vis Sci. 2013. PMID: 23737474
-
Antagonism between Gdf6a and retinoic acid pathways controls timing of retinal neurogenesis and growth of the eye in zebrafish.Development. 2016 Apr 1;143(7):1087-98. doi: 10.1242/dev.130922. Epub 2016 Feb 18. Development. 2016. PMID: 26893342 Free PMC article.
-
Abnormal Cone and Rod Photoreceptor Morphogenesis in gdf6a Mutant Zebrafish.Invest Ophthalmol Vis Sci. 2020 Apr 9;61(4):9. doi: 10.1167/iovs.61.4.9. Invest Ophthalmol Vis Sci. 2020. PMID: 32293666 Free PMC article.
-
Looking to the future of zebrafish as a model to understand the genetic basis of eye disease.Hum Genet. 2019 Sep;138(8-9):993-1000. doi: 10.1007/s00439-019-02055-z. Epub 2019 Aug 17. Hum Genet. 2019. PMID: 31422478 Free PMC article. Review.
-
Animal and cellular models of microphthalmia.Ther Adv Rare Dis. 2021 Feb 27;2:2633004021997447. doi: 10.1177/2633004021997447. eCollection 2021 Jan-Dec. Ther Adv Rare Dis. 2021. PMID: 37181112 Free PMC article. Review.
Cited by
-
The dynamic role of bone morphogenetic proteins in neural stem cell fate and maturation.Dev Neurobiol. 2012 Jul;72(7):1068-84. doi: 10.1002/dneu.22022. Dev Neurobiol. 2012. PMID: 22489086 Free PMC article. Review.
-
The master male sex determinant Gdf6Y of the turquoise killifish arose through allelic neofunctionalization.Nat Commun. 2025 Jan 9;16(1):540. doi: 10.1038/s41467-025-55899-7. Nat Commun. 2025. PMID: 39788971 Free PMC article.
-
Role of Thyroid Hormone in Dynamic Variation of gdf6a Gene during Metamorphosis of Paralichthys olivaceus.Int J Mol Sci. 2023 Dec 19;25(1):23. doi: 10.3390/ijms25010023. Int J Mol Sci. 2023. PMID: 38203198 Free PMC article.
-
GDF6-CD99 Signaling Regulates Src and Ewing Sarcoma Growth.Cell Rep. 2020 Nov 3;33(5):108332. doi: 10.1016/j.celrep.2020.108332. Cell Rep. 2020. PMID: 33147457 Free PMC article.
-
Association of rs6982567 near GDF6 with neovascular age-related macular degeneration and polypoidal choroidal vasculopathy in a Han Chinese cohort.BMC Ophthalmol. 2014 Nov 22;14:140. doi: 10.1186/1471-2415-14-140. BMC Ophthalmol. 2014. PMID: 25416513 Free PMC article.
References
-
- Lowry RB, Kohut R, Sibbald B, Rouleau J. Anophthalmia and microphthalmia in the Alberta Congenital Anomalies Surveillance System. Can J Ophthalmol. 2005;40:38–44. - PubMed
-
- Shaw GM, Carmichael SL, Yang W, Harris JA, Finnell RH, Lammer EJ. Epidemiologic characteristics of anophthalmia and bilateral microphthalmia among 2.5 million births in California, 1989-1997. Am J Med Genet A. 2005;137:36–40. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

