The structure and allosteric regulation of glutamate dehydrogenase
- PMID: 21070828
- PMCID: PMC3135769
- DOI: 10.1016/j.neuint.2010.10.017
The structure and allosteric regulation of glutamate dehydrogenase
Abstract
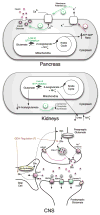
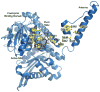
Glutamate dehydrogenase (GDH) has been extensively studied for more than 50 years. Of particular interest is the fact that, while considered by most to be a 'housekeeping' enzyme, the animal form of GDH is heavily regulated by a wide array of allosteric effectors and exhibits extensive inter-subunit communication. While the chemical mechanism for GDH has remained unchanged through epochs of evolution, it was not clear how or why animals needed to evolve such a finely tuned form of this enzyme. As reviewed here, recent studies have begun to elucidate these issues. Allosteric regulation first appears in the Ciliates and may have arisen to accommodate evolutionary changes in organelle function. The occurrence of allosteric regulation appears to be coincident with the formation of an 'antenna' like feature rising off the tops of the subunits that may be necessary to facilitate regulation. In animals, this regulation further evolved as GDH became integrated into a number of other regulatory pathways. In particular, mutations in GDH that abrogate GTP inhibition result in dangerously high serum levels of insulin and ammonium. Therefore, allosteric regulation of GDH plays an important role in insulin homeostasis. Finally, several compounds have been identified that block GDH-mediated insulin secretion that may be to not only find use in treating these insulin disorders but to kill tumors that require glutamine metabolism for cellular energy.
Copyright © 2010 Elsevier Ltd. All rights reserved.
Figures

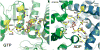

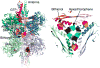
Similar articles
-
Untangling the glutamate dehydrogenase allosteric nightmare.Trends Biochem Sci. 2008 Nov;33(11):557-64. doi: 10.1016/j.tibs.2008.07.007. Epub 2008 Sep 24. Trends Biochem Sci. 2008. PMID: 18819805 Review.
-
Evolution of glutamate dehydrogenase regulation of insulin homeostasis is an example of molecular exaptation.Biochemistry. 2004 Nov 16;43(45):14431-43. doi: 10.1021/bi048817i. Biochemistry. 2004. PMID: 15533048
-
Glutamate dehydrogenase: structure, allosteric regulation, and role in insulin homeostasis.Neurochem Res. 2014;39(3):433-45. doi: 10.1007/s11064-013-1173-2. Epub 2013 Oct 12. Neurochem Res. 2014. PMID: 24122080 Free PMC article. Review.
-
Expression, purification and characterization of human glutamate dehydrogenase (GDH) allosteric regulatory mutations.Biochem J. 2002 Apr 1;363(Pt 1):81-7. doi: 10.1042/0264-6021:3630081. Biochem J. 2002. PMID: 11903050 Free PMC article.
-
The structure and allosteric regulation of mammalian glutamate dehydrogenase.Arch Biochem Biophys. 2012 Mar 15;519(2):69-80. doi: 10.1016/j.abb.2011.10.015. Epub 2011 Nov 4. Arch Biochem Biophys. 2012. PMID: 22079166 Free PMC article. Review.
Cited by
-
Untargeted metabolomics confirms the association between plasma branched chain amino acids and residual feed intake in beef heifers.PLoS One. 2022 Nov 29;17(11):e0277458. doi: 10.1371/journal.pone.0277458. eCollection 2022. PLoS One. 2022. PMID: 36445891 Free PMC article.
-
Very-low-carbohydrate diet enhances human T-cell immunity through immunometabolic reprogramming.EMBO Mol Med. 2021 Aug 9;13(8):e14323. doi: 10.15252/emmm.202114323. Epub 2021 Jun 21. EMBO Mol Med. 2021. PMID: 34151532 Free PMC article.
-
Glutamate dehydrogenase activator BCH stimulating reductive amination prevents high fat/high fructose diet-induced steatohepatitis and hyperglycemia in C57BL/6J mice.Sci Rep. 2016 Nov 22;5:37468. doi: 10.1038/srep37468. Sci Rep. 2016. PMID: 27874078 Free PMC article.
-
Mitochondrial regulation of β-cell function: maintaining the momentum for insulin release.Mol Aspects Med. 2015 Apr;42:91-104. doi: 10.1016/j.mam.2015.01.004. Epub 2015 Feb 7. Mol Aspects Med. 2015. PMID: 25659350 Free PMC article. Review.
-
Structural Studies of Glutamate Dehydrogenase (Isoform 1) From Arabidopsis thaliana, an Important Enzyme at the Branch-Point Between Carbon and Nitrogen Metabolism.Front Plant Sci. 2020 Jun 5;11:754. doi: 10.3389/fpls.2020.00754. eCollection 2020. Front Plant Sci. 2020. PMID: 32655590 Free PMC article.
References
-
- Allen A, Kwagh J, Fang J, Stanley CA, Smith TJ. Evolution of glutamate dehydrogenase regulation of insulin homeostasis is an example of molecular exaptation. Biochemistry. 2004;43:14431–14443. - PubMed
-
- Aubert S, Bligny R, Douce R, Ratcliffe RG, Roberts JKM. Contribution of glutamate dehydrogenase to mitochondrial metabolism studied by 13C and 31P nuclear magnetic resonance. J Exp Bot. 2001;52:37–45. - PubMed
-
- Bahi-Buisson N, Roze E, Dionisi C, Escande F, Valayannopoulos V, Feillet F, Heinrichs C. Neurological aspects of hyperinsulinism-hyperammonaemia syndrome. Dev Med & Child Neurol. 2008;50:945–949. - PubMed
-
- Bailey JS, Bell ET, Bell JE. Regulation of bovine glutamate dehydrogenase. J Biol Chem. 1982;257:5579–5583. - PubMed
-
- Banerjee S, Schmidt T, Fang J, Stanley CA, Smith TJ. Structural studies on ADP activation of mammalian glutamate dehydrogenase and the evolution of regulation. Biochemistry. 2003;42:3446–3456. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

