Paracrine modulation of cholangiocyte serotonin synthesis orchestrates biliary remodeling in adults
- PMID: 21071507
- PMCID: PMC3043647
- DOI: 10.1152/ajpgi.00368.2010
Paracrine modulation of cholangiocyte serotonin synthesis orchestrates biliary remodeling in adults
Abstract
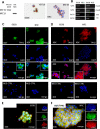
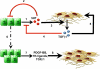
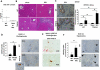
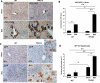
Paracrine signaling between cholangiocytes and stromal cells regulates biliary remodeling. Cholangiocytes have neuroepithelial characteristics and serotonin receptor agonists inhibit their growth, but whether they are capable of serotonin biosynthesis is unknown. We hypothesized that cholangiocytes synthesize serotonin and that cross talk between liver myofibroblasts (MF) and cholangiocytes regulates this process to influence biliary remodeling. Transwell cultures of cholangiocytes ± MF, and tryptophan hydroxylase-2 knockin (TPH2KI) mice with an inactivating mutation of the neuronal tryptophan hydroxylase (TPH) isoform, TPH2, were evaluated. Results in the cell culture models confirm that cholangiocytes have serotonin receptors and demonstrate for the first time that these cells express TPH2 and produce serotonin, which autoinhibits their growth but stimulates MF production of TGF-β(1). Increased TGF-β(1), in turn, counteracts autocrine inhibition of cholangiocyte growth by repressing cholangiocyte TPH2 expression. Studies of TPH2KI mice confirm that TPH2-mediated production of serotonin plays an important role in remodeling damaged bile ducts because mice with decreased TPH2 function have reduced biliary serotonin levels and exhibit excessive cholangiocyte proliferation, accumulation of aberrant ductules and liver progenitors, and increased liver fibrosis after bile duct ligation. This new evidence that cholangiocytes express the so-called neuronal isoform of TPH, synthesize serotonin de novo, and deploy serotonin as an autocrine/paracrine signal to regulate regeneration of the biliary tree complements earlier work that revealed that passive release of serotonin from platelets stimulates hepatocyte proliferation. Given the prevalent use of serotonin-modulating drugs, these findings have potentially important implications for recovery from various types of liver damage.
Figures



Similar articles
-
Autocrine/paracrine regulation of the growth of the biliary tree by the neuroendocrine hormone serotonin.Gastroenterology. 2005 Jan;128(1):121-37. doi: 10.1053/j.gastro.2004.10.002. Gastroenterology. 2005. PMID: 15633129
-
Inhibition of integrin alphavbeta6 on cholangiocytes blocks transforming growth factor-beta activation and retards biliary fibrosis progression.Gastroenterology. 2008 Aug;135(2):660-70. doi: 10.1053/j.gastro.2008.04.009. Epub 2008 Apr 16. Gastroenterology. 2008. PMID: 18538673 Free PMC article.
-
Progesterone stimulates the proliferation of female and male cholangiocytes via autocrine/paracrine mechanisms.Am J Physiol Gastrointest Liver Physiol. 2008 Jul;295(1):G124-G136. doi: 10.1152/ajpgi.00536.2007. Epub 2008 May 29. Am J Physiol Gastrointest Liver Physiol. 2008. Retraction in: Am J Physiol Gastrointest Liver Physiol. 2024 Sep 1;327(3):G481. doi: 10.1152/ajpgi.00536.2007_RET. PMID: 18511743 Free PMC article. Retracted.
-
Mechanisms of cholangiocyte responses to injury.Biochim Biophys Acta Mol Basis Dis. 2018 Apr;1864(4 Pt B):1262-1269. doi: 10.1016/j.bbadis.2017.06.017. Epub 2017 Jun 23. Biochim Biophys Acta Mol Basis Dis. 2018. PMID: 28648950 Free PMC article. Review.
-
Regulators of Cholangiocyte Proliferation.Gene Expr. 2017 Feb 10;17(2):155-171. doi: 10.3727/105221616X692568. Epub 2016 Jul 12. Gene Expr. 2017. PMID: 27412505 Free PMC article. Review.
Cited by
-
Discrete Correlation Summation Clustering Reveals Differential Regulation of Liver Metabolism by Thrombospondin-1 in Low-Fat and High-Fat Diet-Fed Mice.Metabolites. 2022 Oct 28;12(11):1036. doi: 10.3390/metabo12111036. Metabolites. 2022. PMID: 36355119 Free PMC article.
-
Recent advances in understanding bile duct remodeling and fibrosis.F1000Res. 2018 Jul 31;7:F1000 Faculty Rev-1165. doi: 10.12688/f1000research.14578.1. eCollection 2018. F1000Res. 2018. PMID: 30109019 Free PMC article. Review.
-
Evaluation of the serotonin pathway as a biomarker in cholangiocarcinoma.J Gastrointest Oncol. 2023 Oct 31;14(5):2192-2201. doi: 10.21037/jgo-23-115. Epub 2023 Sep 15. J Gastrointest Oncol. 2023. PMID: 37969829 Free PMC article.
-
Functional and structural features of cholangiocytes in health and disease.Cell Mol Gastroenterol Hepatol. 2015 Jul 1;1(4):368-380. doi: 10.1016/j.jcmgh.2015.05.005. Cell Mol Gastroenterol Hepatol. 2015. PMID: 26273695 Free PMC article.
-
Serotonergic Regulation of Hepatic Energy Metabolism.Endocrinol Metab (Seoul). 2021 Dec;36(6):1151-1160. doi: 10.3803/EnM.2021.1331. Epub 2021 Dec 16. Endocrinol Metab (Seoul). 2021. PMID: 34911172 Free PMC article. Review.
References
-
- Dewachter L, Adnot S, Fadel E, Humbert M, Maitre B, Barlier-Mur AM, Simonneau G, Hamon M, Naeije R, Eddahibi S. Angiopoietin/Tie2 pathway influences smooth muscle hyperplasia in idiopathic pulmonary hypertension. Am J Respir Crit Care Med 174: 1025–1033, 2006 - PubMed
-
- Eddahibi S, Guignabert C, Barlier-Mur AM, Dewachter L, Fadel E, Dartevelle P, Humbert M, Simonneau G, Hanoun N, Saurini F, Hamon M, Adnot S. Cross talk between endothelial and smooth muscle cells in pulmonary hypertension: critical role for serotonin-induced smooth muscle hyperplasia. Circulation 113: 1857–1864, 2006 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

