Loss of 'small-world' networks in Alzheimer's disease: graph analysis of FMRI resting-state functional connectivity
- PMID: 21072180
- PMCID: PMC2967467
- DOI: 10.1371/journal.pone.0013788
Loss of 'small-world' networks in Alzheimer's disease: graph analysis of FMRI resting-state functional connectivity
Abstract
Background: Local network connectivity disruptions in Alzheimer's disease patients have been found using graph analysis in BOLD fMRI. Other studies using MEG and cortical thickness measures, however, show more global long distance connectivity changes, both in functional and structural imaging data. The form and role of functional connectivity changes thus remains ambiguous. The current study shows more conclusive data on connectivity changes in early AD using graph analysis on resting-state condition fMRI data.
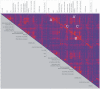
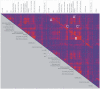
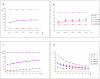
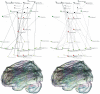
Methodology/principal findings: 18 mild AD patients and 21 healthy age-matched control subjects without memory complaints were investigated in resting-state condition with MRI at 1.5 Tesla. Functional coupling between brain regions was calculated on the basis of pair-wise synchronizations between regional time-series. Local (cluster coefficient) and global (path length) network measures were quantitatively defined. Compared to controls, the characteristic path length of AD functional networks is closer to the theoretical values of random networks, while no significant differences were found in cluster coefficient. The whole-brain average synchronization does not differ between Alzheimer and healthy control groups. Post-hoc analysis of the regional synchronization reveals increased AD synchronization involving the frontal cortices and generalized decreases located at the parietal and occipital regions. This effectively translates in a global reduction of functional long-distance links between frontal and caudal brain regions.
Conclusions/significance: We present evidence of AD-induced changes in global brain functional connectivity specifically affecting long-distance connectivity. This finding is highly relevant for it supports the anterior-posterior disconnection theory and its role in AD. Our results can be interpreted as reflecting the randomization of the brain functional networks in AD, further suggesting a loss of global information integration in disease.
Conflict of interest statement
Figures

Similar articles
-
Identifying patients with Alzheimer's disease using resting-state fMRI and graph theory.Clin Neurophysiol. 2015 Nov;126(11):2132-41. doi: 10.1016/j.clinph.2015.02.060. Epub 2015 Apr 1. Clin Neurophysiol. 2015. PMID: 25907414
-
Functional neural network analysis in frontotemporal dementia and Alzheimer's disease using EEG and graph theory.BMC Neurosci. 2009 Aug 21;10:101. doi: 10.1186/1471-2202-10-101. BMC Neurosci. 2009. PMID: 19698093 Free PMC article.
-
[Structural and functional neuronal connectivity in Alzheimer's disease: a combined DTI and fMRI study].Nervenarzt. 2012 Jul;83(7):878-87. doi: 10.1007/s00115-011-3326-3. Nervenarzt. 2012. PMID: 21713583 German.
-
Binary and weighted network analysis and its applications to functional connectivity in subjective memory complaints: A resting-state fMRI approach.Ageing Res Rev. 2025 Apr;106:102688. doi: 10.1016/j.arr.2025.102688. Epub 2025 Feb 11. Ageing Res Rev. 2025. PMID: 39947486 Review.
-
A survey on applications and analysis methods of functional magnetic resonance imaging for Alzheimer's disease.J Neurosci Methods. 2019 Apr 1;317:121-140. doi: 10.1016/j.jneumeth.2018.12.012. Epub 2018 Dec 26. J Neurosci Methods. 2019. PMID: 30593787 Review.
Cited by
-
Influence of choice of null network on small-world parameters of structural correlation networks.PLoS One. 2013 Jun 28;8(6):e67354. doi: 10.1371/journal.pone.0067354. Print 2013. PLoS One. 2013. PMID: 23840672 Free PMC article.
-
Reorganization of Brain Networks in Aging and Age-related Diseases.Aging Dis. 2012 Apr;3(2):181-93. Epub 2011 Nov 28. Aging Dis. 2012. PMID: 22724079 Free PMC article.
-
Multiscale entropy and small-world network analysis in rs-fMRI - new tools to evaluate early basal ganglia dysfunction in diabetic peripheral neuropathy.Front Endocrinol (Lausanne). 2022 Nov 4;13:974254. doi: 10.3389/fendo.2022.974254. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 36407323 Free PMC article.
-
Molecular and Imaging Biomarkers in Alzheimer's Disease: A Focus on Recent Insights.J Pers Med. 2020 Jul 10;10(3):61. doi: 10.3390/jpm10030061. J Pers Med. 2020. PMID: 32664352 Free PMC article. Review.
-
Functional Brain Networks: Does the Choice of Dependency Estimator and Binarization Method Matter?Sci Rep. 2016 Jul 15;6:29780. doi: 10.1038/srep29780. Sci Rep. 2016. PMID: 27417262 Free PMC article.
References
-
- Delbeuck X, Van der Linden M, Collette F. Alzheimer's disease as a disconnection syndrome? Neuropsychol Rev. 2003;13:79–92. - PubMed
-
- O'Sullivan M, Jones DK, Summers PE, Morris RG, Williams SC, et al. Evidence for cortical “disconnection” as a mechanism of age-related cognitive decline. Neurology. 2001;57:632–638. - PubMed
-
- Desimone R, Duncan J. Neural mechanisms of selective visual attention. Annu Rev Neurosci. 1995;18:193–222. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

