Role of the gp85/trans-sialidases in Trypanosoma cruzi tissue tropism: preferential binding of a conserved peptide motif to the vasculature in vivo
- PMID: 21072227
- PMCID: PMC2970537
- DOI: 10.1371/journal.pntd.0000864
Role of the gp85/trans-sialidases in Trypanosoma cruzi tissue tropism: preferential binding of a conserved peptide motif to the vasculature in vivo
Abstract
Background: Transmitted by blood-sucking insects, the unicellular parasite Trypanosoma cruzi is the causative agent of Chagas' disease, a malady manifested in a variety of symptoms from heart disease to digestive and urinary tract dysfunctions. The reasons for such organ preference have been a matter of great interest in the field, particularly because the parasite can invade nearly every cell line and it can be found in most tissues following an infection. Among the molecular factors that contribute to virulence is a large multigene family of proteins known as gp85/trans-sialidase, which participates in cell attachment and invasion. But whether these proteins also contribute to tissue homing had not yet been investigated. Here, a combination of endothelial cell immortalization and phage display techniques has been used to investigate the role of gp85/trans-sialidase in binding to the vasculature.
Methods: Bacteriophage expressing an important peptide motif (denominated FLY) common to all gp85/trans-sialidase proteins was used as a surrogate to investigate the interaction of this motif with the endothelium compartment. For that purpose phage particles were incubated with endothelial cells obtained from different organs or injected into mice intravenously and the number of phage particles bound to cells or tissues was determined. Binding of phages to intermediate filament proteins has also been studied.
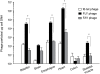
Findings and conclusions: Our data indicate that FLY interacts with the endothelium in an organ-dependent manner with significantly higher avidity for the heart vasculature. Phage display results also show that FLY interaction with intermediate filament proteins is not limited to cytokeratin 18 (CK18), which may explain the wide variety of cells infected by the parasite. This is the first time that members of the intermediate filaments in general, constituted by a large group of ubiquitously expressed proteins, have been implicated in T. cruzi cell invasion and tissue homing.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
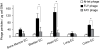
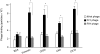
Similar articles
-
Trypanosoma cruzi Binds to Cytokeratin through Conserved Peptide Motifs Found in the Laminin-G-Like Domain of the gp85/Trans-sialidase Proteins.PLoS Negl Trop Dis. 2015 Sep 23;9(9):e0004099. doi: 10.1371/journal.pntd.0004099. eCollection 2015 Sep. PLoS Negl Trop Dis. 2015. PMID: 26398185 Free PMC article.
-
A conserved domain of the gp85/trans-sialidase family activates host cell extracellular signal-regulated kinase and facilitates Trypanosoma cruzi infection.Exp Cell Res. 2007 Jan 1;313(1):210-8. doi: 10.1016/j.yexcr.2006.10.008. Epub 2006 Oct 17. Exp Cell Res. 2007. PMID: 17101128
-
Endothelial cell signalling induced by trans-sialidase from Trypanosoma cruzi.Cell Microbiol. 2008 Jan;10(1):88-99. doi: 10.1111/j.1462-5822.2007.01017.x. Epub 2007 Aug 2. Cell Microbiol. 2008. PMID: 17672865
-
Trans-sialidase, SAPA amino acid repeats and the relationship between Trypanosoma cruzi and the mammalian host.Parasitology. 1994;108 Suppl:S37-44. doi: 10.1017/s0031182000075703. Parasitology. 1994. PMID: 8084653 Review.
-
The Gp85 surface glycoproteins from Trypanosoma cruzi.Subcell Biochem. 2014;74:151-80. doi: 10.1007/978-94-007-7305-9_7. Subcell Biochem. 2014. PMID: 24264245 Review.
Cited by
-
The Recombinant Form of Trypanosoma cruzi P21 Controls Infection by Modulating Host Immune Response.Front Immunol. 2020 Jun 5;11:1010. doi: 10.3389/fimmu.2020.01010. eCollection 2020. Front Immunol. 2020. PMID: 32655546 Free PMC article.
-
The Screen of a Phage Display Library Identifies a Peptide That Binds to the Surface of Trypanosoma cruzi Trypomastigotes and Impairs Their Infection of Mammalian Cells.Front Microbiol. 2022 Mar 10;13:864788. doi: 10.3389/fmicb.2022.864788. eCollection 2022. Front Microbiol. 2022. PMID: 35359712 Free PMC article.
-
Trypanosoma cruzi infection and host lipid metabolism.Mediators Inflamm. 2014;2014:902038. doi: 10.1155/2014/902038. Epub 2014 Sep 3. Mediators Inflamm. 2014. PMID: 25276058 Free PMC article. Review.
-
Thinking outside the blood: Perspectives on tissue-resident Trypanosoma brucei.PLoS Pathog. 2021 Sep 16;17(9):e1009866. doi: 10.1371/journal.ppat.1009866. eCollection 2021 Sep. PLoS Pathog. 2021. PMID: 34529724 Free PMC article. Review.
-
Endothelial transmigration by Trypanosoma cruzi.PLoS One. 2013 Dec 2;8(12):e81187. doi: 10.1371/journal.pone.0081187. eCollection 2013. PLoS One. 2013. PMID: 24312535 Free PMC article. Clinical Trial.
References
-
- Alves M, Colli W. Trypanosoma cruzi: adhesion to the host cell and intracellular survival. IUBMB Life. 2007;59:274–279. - PubMed
-
- Bittencourt AL, Vieira GO, Tavares HC, Mota E, Maguire J. Esophageal involvement in congenital Chagas' disease: Report of a case with megaesophagus. American Journal of Tropical Medicine and Hygiene. 1984;33:30–33. - PubMed
-
- Boczko J, Tar M, Melman A, Jelicks L, Wittner M, et al. Trypanosoma cruzi infection induced changes in the innervation, structure and function of the murine bladder. Journal of Urology. 2005;173:1784–1788. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

