Curcumin activates the p38MPAK-HSP25 pathway in vitro but fails to attenuate diabetic nephropathy in DBA2J mice despite urinary clearance documented by HPLC
- PMID: 21073732
- PMCID: PMC2999583
- DOI: 10.1186/1472-6882-10-67
Curcumin activates the p38MPAK-HSP25 pathway in vitro but fails to attenuate diabetic nephropathy in DBA2J mice despite urinary clearance documented by HPLC
Abstract
Background: Curcumin has anti-inflammatory, anti-oxidant, and anti-proliferative properties, and depending upon the experimental circumstances, may be pro- or anti-apoptotic. Many of these biological actions could ameliorate diabetic nephropathy.
Methods/design: Mouse podocytes, cultured in basal or high glucose conditions, underwent acute exposure to curcumin. Western blots for p38-MAPK, COX-2 and cleaved caspase-3; isoelectric focusing for HSP25 phosphorylation; and DNase I assays for F- to G- actin cleavage were performed for in vitro analyses. In vivo studies examined the effects of dietary curcumin on the development of diabetic nephropathy in streptozotocin (Stz)-induced diabetes in DBA2J mice. Urinary albumin to creatinine ratios were obtained, high performance liquid chromatography was performed for urinary curcuminoid measurements, and Western blots for p38-MAPK and total HSP25 were performed.
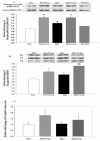
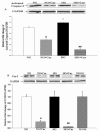
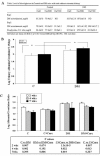
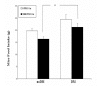
Results: Curcumin enhanced the phosphorylation of both p38MAPK and downstream HSP25; inhibited COX-2; induced a trend towards attenuation of F- to G-actin cleavage; and dramatically inhibited the activation of caspase-3 in vitro. In curcumin-treated DBA2J mice with Stz-diabetes, HPLC measurements confirmed the presence of urinary curcuminoid. Nevertheless, dietary provision of curcumin either before or after the induction of diabetes failed to attenuate albuminuria.
Conclusions: Apart from species, strain, early differences in glycemic control, and/or dosing effects, the failure to modulate albuminuria may have been due to a decrement in renal HSP25 or stimulation of the 12/15 lipoxygenase pathway in DBA2J mice fed curcumin. In addition, these studies suggest that timed urine collections may be useful for monitoring curcumin dosing and renal pharmacodynamic effects.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

