Integration of brassinosteroid signal transduction with the transcription network for plant growth regulation in Arabidopsis
- PMID: 21074725
- PMCID: PMC3018842
- DOI: 10.1016/j.devcel.2010.10.010
Integration of brassinosteroid signal transduction with the transcription network for plant growth regulation in Arabidopsis
Abstract
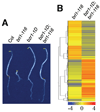
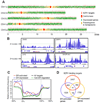
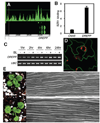
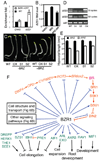
Brassinosteroids (BRs) regulate a wide range of developmental and physiological processes in plants through a receptor-kinase signaling pathway that controls the BZR transcription factors. Here, we use transcript profiling and chromatin-immunoprecipitation microarray (ChIP-chip) experiments to identify 953 BR-regulated BZR1 target (BRBT) genes. Functional studies of selected BRBTs further demonstrate roles in BR promotion of cell elongation. The BRBT genes reveal numerous molecular links between the BR-signaling pathway and downstream components involved in developmental and physiological processes. Furthermore, the results reveal extensive crosstalk between BR and other hormonal and light-signaling pathways at multiple levels. For example, BZR1 not only controls the expression of many signaling components of other hormonal and light pathways but also coregulates common target genes with light-signaling transcription factors. Our results provide a genomic map of steroid hormone actions in plants that reveals a regulatory network that integrates hormonal and light-signaling pathways for plant growth regulation.
Copyright © 2010 Elsevier Inc. All rights reserved.
Figures



References
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

