Betulinic acid inhibits endotoxin-stimulated phosphorylation cascade and pro-inflammatory prostaglandin E(2) production in human peripheral blood mononuclear cells
- PMID: 21077850
- PMCID: PMC3058162
- DOI: 10.1111/j.1476-5381.2010.01112.x
Betulinic acid inhibits endotoxin-stimulated phosphorylation cascade and pro-inflammatory prostaglandin E(2) production in human peripheral blood mononuclear cells
Erratum in
-
Correction.Br J Pharmacol. 2021 Jun;178(12):2548. doi: 10.1111/bph.15519. Br J Pharmacol. 2021. PMID: 34029381 No abstract available.
Abstract
Background and purpose: Betulinic acid (BA) is a naturally occurring triterpenoid widely distributed throughout the plant kingdom. We previously reported that BA inhibits lipopolysaccharide (LPS)-induced interleukin-6 production through modulation of nuclear factor κB (NF-κB) in human peripheral blood mononuclear cells (hPBMCs). This study attempted to identify other mechanisms through which BA modulates LPS signalling in mononuclear cells. The effects of BA on signalling pathways downstream were focused on in this study.
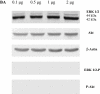
Experimental approach: We determined the ability of BA to interfere with p38 and extracellular regulated kinase (ERK) phosphorylation as well as Akt phosphorylation and nuclear factor-κB activation using LPS-activated hPBMCs as an in vitro model. LPS-induced endotoxin shock in mice was the in vivo model employed.
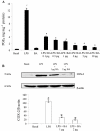
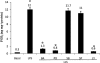
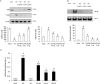
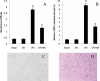
Key results: BA inhibited LPS-induced COX-2 protein expression and prostaglandin E(2) production and also attenuated LPS-induced ERK and Akt phosphorylation, but not p38 in hPBMCs. BA abolished LPS-induced IκBα phosphorylation and thus normalized the levels of IκBα in cytosol. BA also inhibited LPS-induced reactive oxygen species formation and lactate dehydrogenase release. Interestingly, BA improved the life span of mice in endotoxin shock and also inhibited PGE(2) production and myeloperoxidase activity in vivo.
Conclusions and implications: BA modulates LPS-induced COX-2 expression in hPBMCs by inhibiting ERK and Akt pathways as well as by modulating IκBα phosphorylation. At the same time, no cell toxicity was observed. The effect of the drug was confirmed through in vivo experiments. The study gives an insight into the molecular mechanisms of BA.
© 2011 The Authors. British Journal of Pharmacology © 2011 The British Pharmacological Society.
Figures


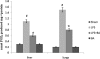
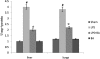

Similar articles
-
Betulinic acid isolated from Bacopa monniera (L.) Wettst suppresses lipopolysaccharide stimulated interleukin-6 production through modulation of nuclear factor-kappaB in peripheral blood mononuclear cells.Int Immunopharmacol. 2010 Aug;10(8):843-9. doi: 10.1016/j.intimp.2010.04.013. Epub 2010 Apr 26. Int Immunopharmacol. 2010. PMID: 20430119
-
Betulinic acid suppresses carcinogen-induced NF-kappa B activation through inhibition of I kappa B alpha kinase and p65 phosphorylation: abrogation of cyclooxygenase-2 and matrix metalloprotease-9.J Immunol. 2003 Sep 15;171(6):3278-86. doi: 10.4049/jimmunol.171.6.3278. J Immunol. 2003. PMID: 12960358
-
Transient activation of EGFR/AKT cell survival pathway and expression of survivin contribute to reduced sensitivity of human melanoma cells to betulinic acid.Int J Oncol. 2005 Sep;27(3):823-30. Int J Oncol. 2005. PMID: 16077934
-
The effect of reactive oxygen species on the synthesis of prostanoids from arachidonic acid.J Physiol Pharmacol. 2013 Aug;64(4):409-21. J Physiol Pharmacol. 2013. PMID: 24101387 Review.
-
Cellular and molecular mechanisms underlying the potential of betulinic acid in cancer prevention and treatment.Phytomedicine. 2024 Sep;132:155858. doi: 10.1016/j.phymed.2024.155858. Epub 2024 Jul 4. Phytomedicine. 2024. PMID: 39053249 Review.
Cited by
-
Secondary Metabolites with Biomedical Applications from Plants of the Sarraceniaceae Family.Int J Mol Sci. 2022 Aug 30;23(17):9877. doi: 10.3390/ijms23179877. Int J Mol Sci. 2022. PMID: 36077275 Free PMC article. Review.
-
A novel rose hip preparation with enhanced anti-inflammatory and chondroprotective effects.Mediators Inflamm. 2014;2014:105710. doi: 10.1155/2014/105710. Epub 2014 Oct 13. Mediators Inflamm. 2014. PMID: 25371599 Free PMC article.
-
Anti-Inflammatory and Anticancer Properties of Birch Bark-Derived Betulin: Recent Developments.Plants (Basel). 2021 Dec 3;10(12):2663. doi: 10.3390/plants10122663. Plants (Basel). 2021. PMID: 34961132 Free PMC article. Review.
-
Chloroquine: A Potential Drug in the COVID-19 Scenario.Trans Indian Natl Acad Eng. 2020;5(2):399-410. doi: 10.1007/s41403-020-00114-w. Epub 2020 Jun 7. Trans Indian Natl Acad Eng. 2020. PMID: 38624400 Free PMC article. Review.
-
A review of herbal therapeutics for the prevention and management of poxvirus infections.Arch Microbiol. 2025 Jun 25;207(8):186. doi: 10.1007/s00203-025-04367-3. Arch Microbiol. 2025. PMID: 40560228 Review.
References
-
- Ajizian SJ, English BK, Meals EA. Specific inhibitors of p38 and extracellular signal-regulated kinase mitogen-activated protein kinase pathways block inducible nitric oxide synthase and tumor necrosis factor accumulation in murine macrophages stimulated with lipopolysaccharide and interferon-γ. J Infect Dis. 1999;179:939–944. - PubMed
-
- Balkwill F, Mantovani A. Inflammation and cancer: back to Virchow? Lancet. 2001;357:539–545. - PubMed
-
- Barnes PJ, Karin M. Nuclear factor-κB: a pivotal transcription factor in chronic inflammatory diseases. N Engl J Med. 1997;336:1066–1071. - PubMed
-
- Bellacosa A, Testa JR, Staal SP, Tsichlis PN. A retroviral oncogene, Akt, encoding a serine-threonine kinase containing an SH2-like region. Science. 1991;254:274–277. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

